What are the causes?
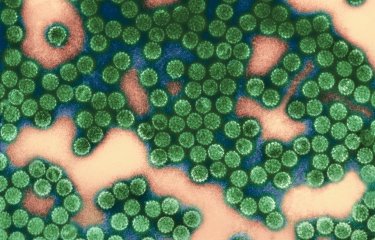
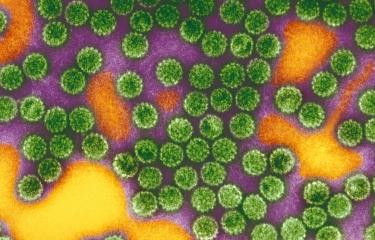
Virtually all cases of cervical cancer (95-100%) are the result of long-term infection with human papillomavirus (HPV). To date, scientists have identified around twenty human papillomaviruses that cause cervical cancer. The two most common are HPV16 and HPV18, which between them are responsible for approximately 70% of cases. 90% of HPV infections are short-lived, and the body's immune defenses manage to eliminate the virus within two years of infection. But in some cases, the infection persists and leads to the development of cancer cells.
It is also important to note that HPV can cause other types of cancer (penile cancer, anal cancer and head and neck cancer).
What are the symptoms?
In the vast majority of cases, HPV infection is asymptomatic. If the infection becomes chronic, it may lead to abnormalities in cervical cells, which can progress to precancerous lesions and then cancer. The time from infection to the development of cancer cells is generally between 5 and 20 years.
Some HPVs cause warts but are not responsible for cancer.
Certain symptoms are associated with cervical cancer, and medical advice should be sought by women experiencing these symptoms, which include vaginal bleeding, lower abdominal pain or pain during sexual intercourse.
How does the virus spread?
Human papillomavirus, which causes cervical cancer, generally spreads during sexual intercourse, with or without penetration. In most cases, infection occurs when a person first becomes sexually active.
How is cervical cancer diagnosed?
Diagnosis is performed via cervical screening (sometimes known as a Pap test or Pap smear), in which a cell sample is collected from the cervix. A cytological examination is then performed under a microscope to identify any abnormalities in the cervical cells. Any precancerous lesions are also detected so that they can be treated before they progress to cancer.
If the result of the cytological examination is positive, additional tests are performed, including a colposcopy (a procedure to examine the cervix in detail) and/or a biopsy.
What treatments are available?
Precancerous lesions are generally treated with surgery. If cancer is found, it is treated with surgery and radiotherapy, supported by chemotherapy. Research is under way to develop a therapeutic vaccine.
How can cervical cancer be prevented?
WHO recommends vaccinating girls in early adolescence. To limit the spread of the virus as much as possible, some countries, including France, recommend vaccinating both girls and boys. Although vaccination reduces the risk of developing cervical cancer threefold, it does not protect against all HPV infection, so it is important for women to undergo regular cervical screening to make sure that any precancerous lesions can be detected and action can be taken before cancer develops.
The use of condoms during sexual intercourse reduces the risk of HPV infection.
How many people are affected?
80% of adults are infected with HPV during their lifetime.
The HPVs that affect genital mucosa occur frequently from the beginning of sexual activity – they are found in a third of women between adolescence and their early twenties. They commonly cause infection, but this usually clears up on its own without any clinical signs. However, infection persists in 3 to 10% of infected women, leading to the risk of developing cervical cancer.
90% of deaths occur in low- and middle-income countries, where access to protection, diagnosis and treatment is more limited.
More than one in five men worldwide are also estimated to be carriers of high-risk HPV.
October 2023