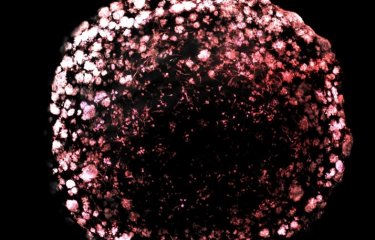
Fifteen researchers from around the world have come to work at Sandrine Etienne-Manneville’s laboratory at the Institut Pasteur. Their brief is to study astrocytes, star-shaped brain cells that support neurons. These cells can become cancerous, causing severe brain tumors known as gliomas, which are the primary cause of childhood cancer mortality ahead of leukemia, with 3,000 new cases diagnosed in France every year. New approaches for tackling these extremely severe tumors could emerge from research carried out by the Cell Polarity, Migration and Cancer Unit. We went to meet this international team.
The researchers’ chief focus is astrocyte migration. In cases of glioblastoma, which is the most severe type of glioma, tumor astrocytes are able to invade the brain. These “invasive” tumors are extremely difficult to treat, since although it is possible to surgically resect the main tumor, cancer cells that have migrated away from it are inaccessible. So how and why do these cells migrate? This is one of the key questions occupying researchers in the unit. Many of them are specifically involved in examining changes to the cells’ skeleton (cytoskeleton), a network of microtubules, actin filaments, and intermediate filaments that give cells their shape and orientation (or polarity) and influence their migration.
A little fish helping to improve tumor diagnosis

After a 5-year post-doctoral fellowship at the Francis Crick Institute in London, Florent Peglion is going back to his roots to rejoin the lab where he completed his PhD on “the analysis of molecular mechanisms responsible for glioblastoma invasion” in 2012. In order to gain a clear understanding of these forms of cancer, Florent spent 6 months in a neuro-oncology group at Pitié-Salpêtrière Hospital in Paris where he was in contact with neurosurgeons and anatomic pathologists. “One thing I discovered while studying for my PhD was that cells’ “adherens junctions” – which are like Velcro strips in their membranes keeping them tightly bound together – were much less present in tumor astrocytes, thus facilitating their migration”, he explains. After his time in London studying the development of a small nematode worm, he has now changed models to focus on the zebrafish. “This small freshwater fish native to the Ganges offers two benefits. Firstly, its larvae are transparent allowing us to observe cell migration, and secondly its brain contains the same cell types as seen in humans. We can inject the larvae with glioblastoma cells sourced from patients by clinician partners and observe tumor cell migration in a physiological environment. Our aim is to identify biomarkers that could be used to characterize tumors, determine whether or not they are invasive with a view to predicting their aggressiveness, and ultimately improve diagnosis for patients.” This model could also be used in future to select therapeutic molecules by simply adding drug candidates to the aquarium water to identify any inhibitors capable of blocking cancer cell migration, and thus preventing tumor invasion. This technique could be used to test approximately one hundred molecules per day. Florent, who is hoping that his post-doctoral fellowship will lead to a permanent position, views work on potential clinical applications as an essential factor in ensuring that his research is “worthwhile”.

Our aim is to identify biomarkers that could be used to characterize tumors, determine whether or not they are invasive with a view to predicting their aggressiveness, and ultimately improve diagnosis for patients
Florent PeglioResearcher in the Cell Polarity, Migration and Cancer Unit
Monday lab meetings
A lab meeting is held every Monday at 2pm. This week's meeting provides Shailaja Seetharaman, a third-year PhD student from India, with an opportunity to present her latest work to team members and share the results of her experiments. English, the “scientific” language used in international publications and conferences, is mainly used in this lab hosting young researchers and engineers from Italy, India, Sweden, the United Kingdom and Australia, a reflection of the Institut Pasteur's cosmopolitan spirit, with 66 nationalities represented last year.
Shailaja examines microtubules, which form part of the cytoskeleton. Before the cell moves, these all align in the same direction, which is known as polarization. The entire cytoskeleton then retracts at the rear pole, allowing the cell to move in a similar way to a slug. Shailaja observes how astrocytes behave on different substrates, since polarization and migration differ depending on surface rigidity. She also investigates the effect of various proteins that have the potential to reorganize the cytoskeleton and affect cell traction and movement. Finally, she presents images showing microscopic observations of microtubules within cells. Lab leader, Sandrine advises the young PhD student to increase the contrast on the videos so that the microtubules can be more clearly observed. Shailaja highlights the limitations of the microscopy techniques used. “We need the Titan”, she concludes, referring to the ultra-powerful microscope recently installed at the Institut Pasteur, through which specimens can be observed at atomic level.

Astrocytes – the neurons’ “babysitters”
Astrocytes are the main cells in the “macroglia” tissue which supports the central nervous system (see p.). They perform a wide variety of roles, notably enabling the neurons to develop and survive, providing them, for instance, with nutrients such as lactate, which they produce from glucose obtained from the blood vessels, and ensuring an optimal environment for the neurons by “cleaning” the space between cells. Astrocytes also play a role in forming and regulating synapses, which are the connections between neurons. Like neurons, they are structured as a network. Under normal circumstances, adults' astrocytes are immobile and do not divide. However, in most brain diseases with inflammatory components, these astrocytes change shape (polarized morphology) and migrate to the inflammatory site.
Just back from a conference
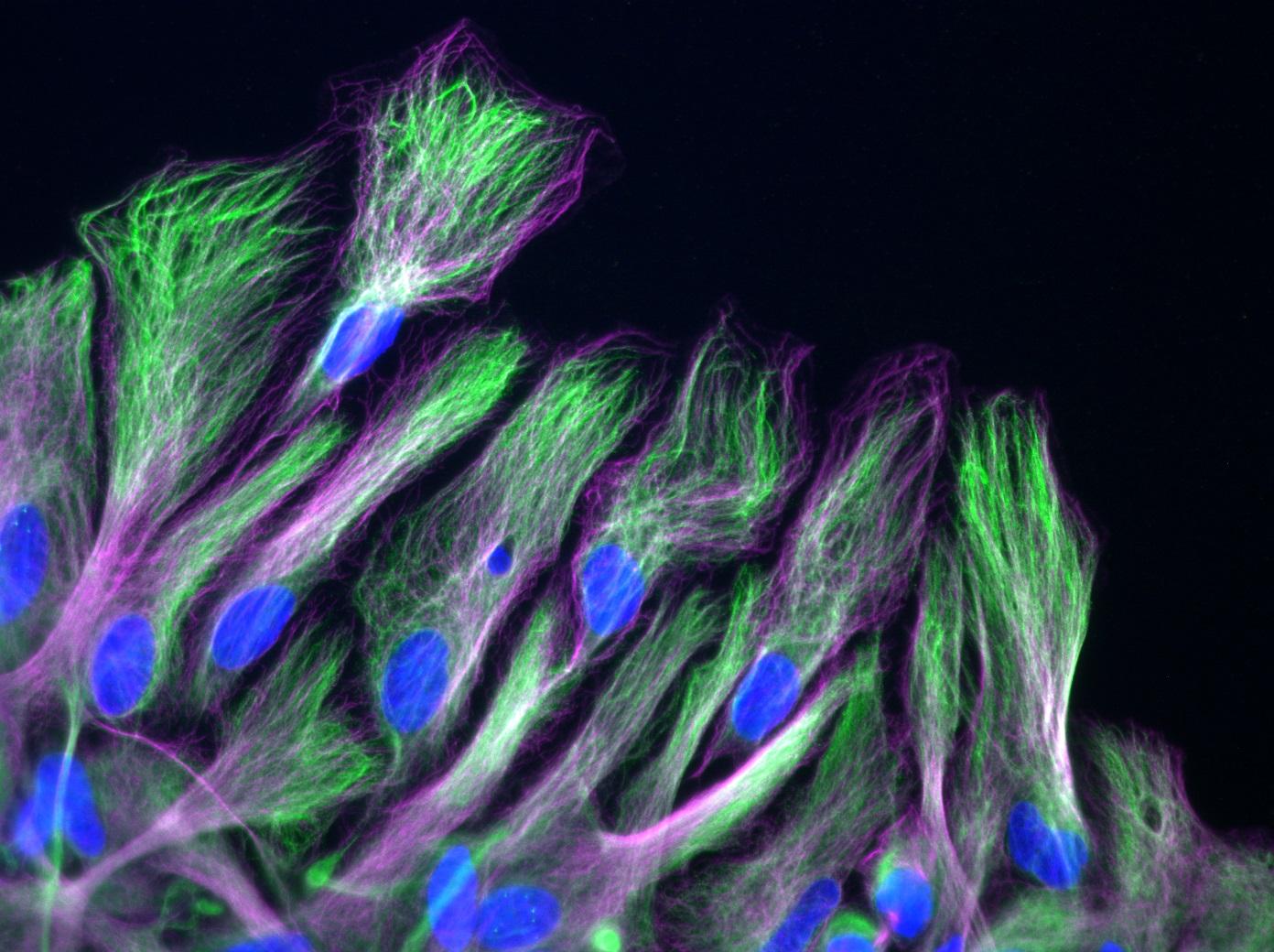
When we meet the fourth-year Italian PhD student Lavinia Capuana who is due to defend her thesis this autumn, she is jet-lagged. Just back from a conference of global cell migration experts in Galveston, Texas, which she attended with Shailaja and Sandrine, she has opted to readjust to the change in time zones by immediately hitting the lab for a busy day straight after her night flight. She speaks enthusiastically about the conference, a rewarding week that enabled her to “discover new techniques, consider partnerships, and find out about other research topics”.
A few days later, she takes us down to the basement of the building, where an aquarium containing zebrafish, an animal model increasingly valued by researchers (see insert on Florent), is set up. Using time-lapse microscopy, it is possible to view and film migrating cells since the fish larvae are transparent. Lavinia is researching a protein involved in collective migration, which she observes in cells forming the aortic vein during the fish’s embryonic development. A deficiency of this protein is observed in 70% of glioma cases, which might explain why tumor astrocytes separate from neighboring cells, migrating individually and at greater speed.
Gliomas – cells that are out of control

Batiste Boeda has been fascinated by the cytoskeleton's “aesthetics” since studying for his PhD at an Institut Pasteur lab specializing in the unrelated field of deafness. Following a spell at a cancer research institute in the UK, he joined Sandrine Etienne-Manneville's team 10 years ago when she was head of a small group for junior researchers (see interview). “Here, we examine polarity in the context of cell migration – in order to migrate, cells must form a front and rear pole, which involves rearranging the cytoskeleton”, he explains. “In adults, astrocytes do not normally move and have no specific polarity. However, they are constantly ready for action – if a cranial injury occurs, they migrate to the scar area to create a “glial” scar. Like firefighters, they are the first response unit to reach the scene. To achieve this, the astrocytes polarize as a “battalion” all marching in the same direction. Migration is orderly and synchronous. In contrast, cancer cells exhibit anarchic migratory behavior, shooting off in different directions. They lose their star shape and become spherical, ricocheting inside the skull like a bullet in a room. This chaotic migration causes damage and ultimately prevents the brain from functioning normally.” The polarity of cancer cells goes awry, hence their anarchic migration. Batiste is specifically investigating Scribble, a protein that regulates polarity. “Instead of forming regular hexagons arranged in a “chicken wire” pattern, Scribble-deficient cells are flat as pancakes and mixed together, one on top of the other”. The Scribble protein interacts with around a hundred other proteins. “We are still just identifying these!” Everyone in the lab works on related topics. Batiste is supervising Astrid, a young Swedish student at the University of Glasgow in the UK, who has come to spend 10 months with the team to research a protein (Cdc42) that is activated by Scribble and acts “as a polarity switch”. “Our main priority is to understand all this complexity before considering therapies.”, emphasizes Batiste. “However, each new stone we turn over potentially has a surprise in store.”
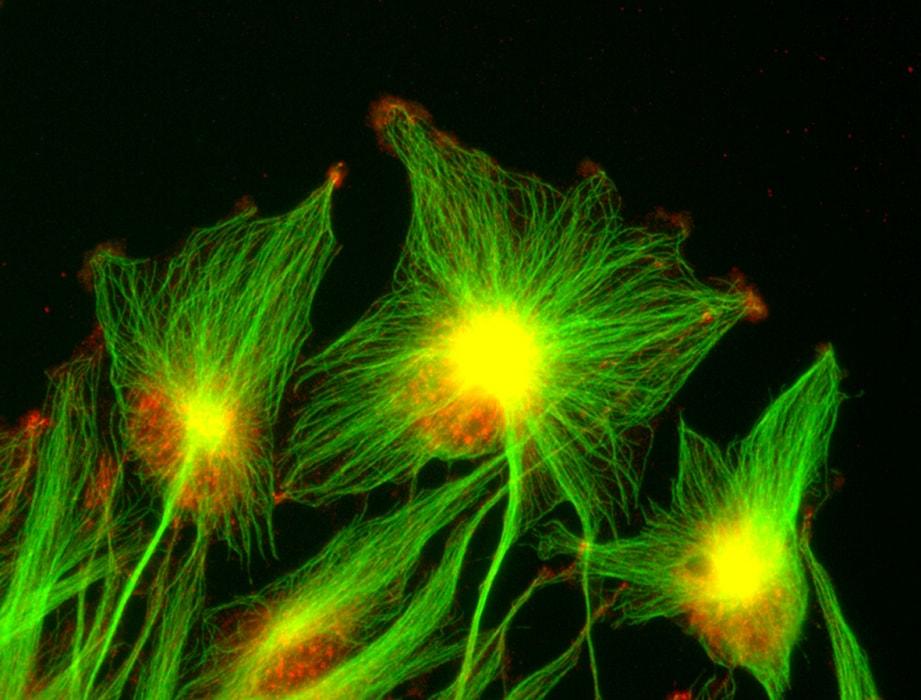
Zebrafish egg laying
Besides reading scientific articles and writing papers, the researchers devote some of their everyday working lives to very practical tasks. For instance, twice a week Lavinia prepares the small fish for egg laying. Using a test tube, she transfers them from their culture tank to a transparent container with two compartments – one for males and the other for females – separated by a removable partition, with a layer of glass beads covering the container bottom. Before embarking on her PhD, Lavinia was introduced to research on zebrafish through an internship at the lab run by Philippe Herbomel, an Institut Pasteur specialist in this model with whom the team collaborates. Based on her practical experience she is able to distinguish between males and females at a glance. The aquarium is kept in darkness overnight until 9am the next day when the lights are turned back on and Lavinia removes the partition separating the males and females. Excited by the light, the males stimulate the females causing them to lay their eggs and then release their sperm, fertilizing the eggs that have fallen to the bottom of the container. These are recovered quickly by rinsing the beads, since Lavinia begins her experiments at the single cell stage.

Precious microliters
Back in the lab on the 3rd floor, everyone has their own area of “bench” – the term affectionately used to describe the surfaces on which experiments are performed. Batiste Boeda (see insert) has just removed an “extremely precious” tube from ice and placed it in a -20°C freezer for safekeeping. “These are antibodies – specific molecules of a given protein, which, when combined with a fluorochrome, are used to ‘label’ this protein” (viewed using a fluorescence microscope). The small tubes, containing no more than 100 microliters of antibodies, cost €400 each, so it is vital not to waste them! Batiste collects “his” cells from his dedicated space in a 37°C incubator where petri dishes of astrocyte cultures are stacked above labels stating their owners’ names: Cécile, Florent, Juliana, etc. He transfers them to small slides similar in appearance to contact lenses. These are treated with a “detergent” to ensure that the cell membranes are permeable and then placed on a drop containing those precious antibodies, which penetrate the cells and "home in" to bind with the protein to be labeled. The slides are then ready to be observed under the microscope either in the lab or at the imaging facility situated in another building on campus (see insert on Cécile).
Every day, each team member follows this process from lab bench to microscope, adding their respective pieces to the puzzle. Driven by their passion for research and a desire to understand, students and experienced researchers alike are aware that their questions concerning glioma cells raise expectations of a breakthrough for patients. Although the research may seem to take a long time, each experiment has the potential to reveal new avenues that will help tackle these diseases in future.

“The microscope lady”
As a child, Cécile Leduc dreamed of becoming a doctor but could not stand the sight of blood. Having initially studied physics, her subsequent career reflects an increasing desire to embrace life sciences with a biology PhD earned in a physical chemistry lab at the Institut Curie, a post-doctoral fellowship on the biophysical properties of motor proteins at the Max Planck Institute in Dresden, Germany, and research at the Institut d’Optique in Bordeaux where she developed optics systems for biological applications. “I moved away from pure physics as it is important for me to see potential benefits for patients, even if these only emerge in the long term”, says the biophysicist who joined Sandrine Etienne-Manneville’s team in 2013. Besides researching “intermediate filaments” – little-known cell skeleton components often over-expressed in cancer patients – Cécile is also in charge of the lab's microscopes. She takes us into a windowless room with four microscopy stations. These are separated by blackout curtains, since observations are performed in darkness due to the fluorescence techniques used. One microscope is used for microinjections, two others for observing individual proteins, and the fourth is referred to by Cécile as the “live microscope”. It is equipped with an incubator, which is a transparent box kept at 37°C, and used to observe migrating living cells, which are filmed by a camera linked up to the microscope. This high-tech equipment purchased a few months ago cost almost €200,000. The incubator alone accounted for €10,000 of this sum, while the camera cost €30,000. If greater accuracy is required for viewing cell components, the researchers use even more powerful (and more expensive!) microscopes at an imaging facility* in another building on campus. “I work with scientists at the facility to develop super-resolution techniques that improve the level of detail. This enables us to view these intermediate filaments, whose role we are seeking to understand, ten times more clearly.”

I moved away from pure physics as it is important for me to see potential benefits for patients, even if these only emerge in the long term
Cécile LeducResearcher in the Cell Polarity, Migration and Cancer Unit

I have been attempting to understand how cells actively and deliberately move from point A to point B
Sandrine Etienne-Manneville is a graduate of the Ecole Normale Supérieure in Paris and has a PhD in Immunology from the University of Paris 6, awarded in 1998. Her laboratory experience includes working at the Cochin Institute of Molecular Genetics during her PhD research, at the University College of London as a post-doctoral fellow, and subsequently at the Institut Curie as a researcher. In 2006, she formed and led the CNRS-affiliated "Cell Polarity and Migration" five-year group at the Institut Pasteur which became a research unit in 2011. Sandrine has also taught at the Ecole Polytechnique (Palaiseau) since 2006, the year in which she was awarded the CNRS Bronze Medal. She has since won many scientific awards and was made a Knight of the National Order of Merit in 2015.
How did you come to work at the Institut Pasteur?
While I was at the Institut Curie, where I was already working on astrocyte migration and polarity, I submitted an application in response to an Institut Pasteur call for proposals on setting up teams of young researchers known as “five-year groups” (G5). My proposal was accepted and I formed a small team in early 2006. I came with a student who I was supervising at the Institut Curie and recruited a technician who has since become an engineer, two post-doctoral fellows, and a PhD student. Five years later in 2011, we were given a positive assessment and the group became a research unit, which allowed us to expand the team. There are a dozen of us now.
What changes were involved in becoming a team leader?
It was something I wanted without necessarily realizing what it involved. In scientific terms, it is obviously a very appealing proposition, since faster progress on research is possible with a team. It is also fascinating from a human perspective. However, I spend a lot of time on administrative tasks, preparing funding applications, completing activity reports, resolving human resources issues, and putting together files for ethics committees. Moreover, special subsidies for young teams were discontinued once we switched from a G5 to a research unit. We are now faced with stiffer funding and scientific competition.
How would you summarize your research topic?
For several years, I have been attempting to understand how cells actively and deliberately move from point A to point B. This phenomenon is essential during embryonic development, when most cells migrate to form tissue and organs, but also in adults. Although we are not aware of it, there is constant movement in our body. Tissue such as the skin and intestinal epithelium is permanently being replaced, wounds close up due to cell multiplication and migration, and in cancer cases, tumor cells migrate to invade tissue or form metastases. Our focus is on astrocytes, cells that normally do not migrate in adults, but can cause glioblastomas, the most invasive of all tumors, which are extremely difficult to treat.
What applications may be anticipated?
A large part of our research is primarily aimed at understanding the mechanisms involved in astrocyte migration and how these cells are affected during glioma formation. Even if it enables new therapeutic targets to be identified, this is basically fundamental research. We have embarked on more applied research on identifying prognostic markers for tumors based on the development of a zebrafish model, which we plan to adapt in future to test drug candidates.
Budding researchers
PhD students, referred to familiarly as “thésards” in French, work three or four years in a lab under a supervisor to develop their research topic before defending their thesis to a panel. Post-doctoral fellows hold a PhD and generally sign a two to five-year fixed-term employment contract. In many cases, several “post-doc” fellowships are needed before landing a permanent position. On January 31, 2019, there were 143 PhD students and 222 post-doctoral fellows in total at all the Institut Pasteur's labs.