Two recently published studies by Institut Pasteur researchers and collaborators shed light onto the ecology and evolution of the bacterial pathogen Listeria monocytogenes. These publications highlight the high prevalence of faecal carriage of the pathogen in asymptomatic hosts, its dependence on the microbiota, and the global spread of its main clinical clone.
Listeria monocytogenes is a major human foodborne pathogen, associated with one of the highest medical and economic burdens. In addition, L. monocytogenes is a well-established model microorganism to study host-pathogen interactions and the pathophysiology of infectious diseases. However, despite its public health and scientific importance, comprehensive studies on the geographic distribution of L. monocytogenes genomic diversity (phylogeography) and the factors that drove its emergence and spread were lacking. Two new studies from Institut Pasteur in collaboration with Université de Paris, the Necker-Enfants malades AP-HP Hospital and international teams provide a better understanding on Listeria ecological niches and the global spread of its main clinical clone.
Identifying the ecological niches of L. monocytogenes
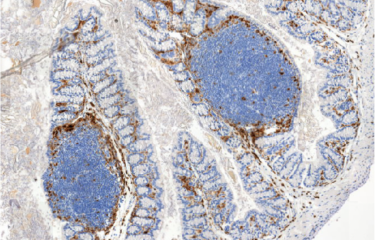
In a study published in Nature Communications, the Biology of Infection Unit at Institut Pasteur and Inserm, together with colleagues from the Universities of Lausanne and Poitiers, studied the distribution of Listeria species by harnessing more than 10,000 metagenomic datasets. The scientists have shown that L. monocytogenes is the most prevalent Listeria species in those datasets, mostly associated with diverse hosts, including humans, cattle and other farm animals, and is also present in soil. While L. monocytogenes is associated to hosts, other non-virulent Listeria species are virtually absent from host datasets.
They further show that L. monocytogenes faecal carriage is common in an independent cohort of close to 1000 faecal samples from asymptomatic donors. This suggests that L. monocytogenes pathogenicity is involved in Listeria host-association, even in the absence of symptoms. By further analyzing the metagenomic datasets, they show that L. monocytogenes faecal carriage is associated with a microbiota signature. This signature is shown to precede L. monocytogenes colonization in an in vivo mouse model, demonstrating that L. monocytogenes faecal carriage depends on the gut microbiota composition. These results indicate that L. monocytogenes faecal carriage is a crucial yet overlooked consequence of its virulence.
Reconstructing the history of the spread of the pathogen
Another study, published in Science advances, highlights the importance of host-association for the global spread of L. monocytogenes. It reports on the first phylogeographic study on L. monocytogenes, focusing on the clinically most prevalent hypervirulent clonal group in Western countries, CC1. By using about 2,000 genomes originating from 40 countries, the Biology of Infection Unit at Institut Pasteur and Inserm, in collaboration with the Biodiversity and Epidemiology of Bacterial Pathogens Unit and the Mathematical Modelling of Infectious Diseases Unit at Institut Pasteur and an international consortium, retraced the evolutionary history of this clone.
The study estimates that main clinical clone CC1 originated in North America and spread worldwide following the Industrial Revolution, through two waves of expansion. This spreading coincided with the expansion of transatlantic meat and livestock trade in the second half of the 19th century, and the rapid growth of cattle farming and food chains in the 20th century. Interestingly, in sharp contrast to its global spread over the past century, recent transmissions chains are mostly local, in farm environments and food production plants, and tend to persist for long periods with limited inter-country spread.
Together these studies provide a comprehensive insight into L. monocytogenes reservoirs and transmission dynamics and highlight the central role of cattle in the spread and evolution of this major foodborne pathogen. These findings also highlight the need to reinforce animal surveillance to reduce the transmission of L. monocytogenes to human through the food chain.
To find out more:
CNR Listeria (in French)
WHO Collaborating Centre : on Pasteur.fr (in French) / on the WHO website (in English)
This study is part of the priority scientific area Emerging infectious diseases of the Institut Pasteur's strategic plan for 2019-2023.
Sources:
Listeria monocytogenes faecal carriage is common and depends on the gut microbiota, Nature Communications, November 24, 2021
Lukas Hafner1, Maxime Pichon2,3,, Christophe Burucoa2,3,, Sophie H. A. Nusser1, Alexandra Moura1,4, Marc Garcia-Garcera5 and Marc Lecuit1,4,6
1 - Institut Pasteur, Université de Paris, Inserm U1117, Biology of Infection Unit, 75015 Paris, France.
2 - University Hospital of Poitiers, Infectious Agents Department, Bacteriology and Infection Control Laboratory, 86021 Poitiers, France.
3 - Université de Poitiers, Faculté de Médecine et de Pharmacie, EA 4331, 86022 Poitiers, France.
4 - Institut Pasteur, National Reference Center and WHO Collaborating Center Listeria, 75015 Paris, France.
5 - University of Lausanne, Department of Fundamental Microbiology, 1015 Lausanne, Switzerland.
6 - Necker-Enfants Malades University Hospital, Division of Infectious Diseases and Tropical Medicine, APHP, Institut Imagine, 75006 Paris, France.
Emergence and global spread of Listeria monocytogenes main clinical clonal complex, Science Advances, December 1st, 2021
Alexandra Moura1,2, Noémie Lefrancq3, Thierry Wirth4,5, Alexandre Leclercq1,2, Vítor Borges6, Brent Gilpin7, Timothy J. Dallman8, Joachim Frey9, Eelco Franz10, Eva M. Nielsen11, Juno Thomas12, Arthur Pightling13, Benjamin P. Howden14,15, Cheryl L. Tarr16, Peter Gerner-Smidt16, Simon Cauchemez3, Henrik Salje3, Sylvain Brisse17, Marc Lecuit1,2,18
1 - Institut Pasteur, Université de Paris, Inserm U1117, Biology of Infection Unit, Paris, France.
2 - Institut Pasteur, National Reference Center and WHO Collaborating Center Listeria, 75015 Paris, France.
3 - Institut Pasteur, Université de Paris, Mathematical Modelling of Infectious Diseases Unit, CNRS UMR 2000, Paris, France.
4 - Institut Systématique Evolution Biodiversité (ISYEB),Museum National d’Histoire Naturelle,
CNRS, Sorbonne Université, Université des Antilles, EPHE, Paris, France.
5 - PSL University, EPHE, Paris, France.
6 - Department of Infectious Diseases, National Institute of Health Dr. Ricardo Jorge, Lisbon, Portugal.
7 - Christchurch Science Centre, Institute of Environmental Science and Research Limited, Christchurch, New Zealand.
8 - Public Health England, London, UK.
9 - Vetsuisse Faculty, University of Bern, Bern, Switzerland.
10 - Centre for Infectious Disease Control, National Institute for Public Health and the Environment (RIVM), Bilthoven, Netherlands.
11 - Statens Serum Institut, Copenhagen, Denmark.
12 - Division of the National Health Laboratory Service, National Institute for Communicable Diseases, Johannesburg, South Africa.
13 - Biostatistics and Bioinformatics, Center for Food Safety and Applied Nutrition, U.S. Food and Drug Administration, College Park, MD, USA.
14 - Microbiological Diagnostic Unit Public Health Laboratory, Department of Microbiology and Immunology, The Doherty Institute for Infection ,and Immunity, University of Melbourne, Melbourne, Victoria, Australia.
15 - Infectious Diseases Department, Austin Health, Heidelberg, Victoria, Australia.
16 - Centers for Disease Control and Prevention, Atlanta, GA, USA.
17 - Institut Pasteur,Université de Paris, Biodiversity and Epidemiology of Bacterial Pathogens Unit, Paris, France.
18 - Necker-Enfants Malades University Hospital, Division of Infectious Diseases and Tropical Medicine, APHP, Institut Imagine, Paris, France.