Rare patients infected by HIV spontaneously control viral replication without antiretroviral therapy, and do not go on to develop AIDS. The ability of these patients, known as "HIV controllers", to limit HIV replication appears to result from a highly effective immune response. Scientists from the Pasteur Institute and Inserm, together with colleagues from Monash University, Melbourne, found that the CD4+ T cells of HIV controllers are capable of recognizing minute amounts of virus. This highly sensitive response is dependent on the expression of specific T cell receptors (TCRs) at the surface of these CD4+ T cells. Scientists determined the crystal structure of TCRs shared by HIV controllers, to understand how they could efficiently recognize a viral antigen and kill HIV-infected cells in persons of different genetic backgrounds. These findings, published in Science Immunology, bring new arguments in the development of cell therapy against HIV.
The existence of a minority of patients who spontaneously control HIV infection provide proof that the human immune system can in rare cases resist the destructive effects of HIV. Researchers are trying to understand the basis of this resistance, to apply this knowledge to the development of vaccines and immunotherapies directed at HIV.
The team of Lisa Chakrabarti (Virus and immunity unit at the Institut Pasteur / Inserm unit U1108), together with the team of Stephanie Gras at Monash University, studied antiviral responses in patients recruited through the French HIV controller cohort ANRS CODEX CO21 coordinated by Olivier Lambotte (Bicêtre hospital). The researchers found that HIV controllers retained antiviral CD4 T cells able to react to minute amounts of virus, while the CD4+ T cells of treated were less efficient at detecting the virus. The sensitive response of controller CD4+ T cells was due to the expression of particular T cell receptors (TCRs) on their surface. These receptors are usually extremely diverse, but the researchers found that identical TCRs could be detected in multiple HIV controllers with different genetic backgrounds.
TCRs recognizes virus fragments (or antigens) bound to specialized molecule of HLA. HLA molecules are like fingerprints: every person has a specific combination of HLA molecules, which help the immune system recognize foreign invaders like bacteria or viruses. In this study, the researchers determined the crystal structure of the TCRs from controllers, and found that these TCRs could recognize a conserved HIV antigen presented by multiple HLA molecules, explaining how the same TCRs could function in genetically diverse HIV controllers.
The researchers then expressed the TCRs from controllers in CD4+ T cells from healthy donors, and found that the simple transfer of a controller TCR was sufficient to confer the capacity to kill HIV-infected cells with high efficiency. CD4+ T cells are usually considered as the cells in charge of helping other effectors such as the killer CD8+ T cells and the antibody-producing B cells. However, when expressing a high-affinity TCRs like those found in controllers, CD4+ T cells become killers themselves, suggesting that they could directly contribute to HIV control. The TCRs of highest affinity could also function when transferred to CD8+ T cells, making them capable of activating both arms of the T cell response.
TCRs from HIV controllers have a series of properties that make them interesting tools for HIV immunotherapy because they can confer the capacity to kill HIV-infected cells to both CD4+ and CD8+ T cells, and they can function in individuals of different genetic backgrounds.

Lisa Chakrabarti, Virus and immunity unit at the Institut Pasteur / Inserm unit U1108, lead author of the study.
TCR transfer therapy could be developed for HIV-infected patients who fail to reconstitute their pool of CD4+ T cells in spite of antiretroviral therapy, and for whom few therapeutic options are available. The objective would be to transfer TCR-modified T cells to these patients, to reconstitute efficient anti-HIV responses, and possibly to interrupt a life-long antiretroviral therapy.

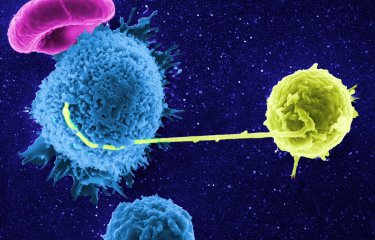
Artistic representation of CD4 + T cells in HIV controllers. © Vanette Tran
These cells are capable of recognizing the same HIV antigen presented by 5 different HLA molecules (HLA-DR1, -DR5, -DR7, -DR11, and -DR15) and killing the thus recognized infected cells.
Source
CD4+ T cell–mediated HLA class II cross-restriction in HIV controllers, Science Immunology, June 8, 2018
Moran Galperin (1*), Carine Farenc (2*), Madhura Mukhopadhyay (1), Dhilshan Jayasinghe (2), Amandine Decroos (1), Daniela Benati (1), Li Lynn Tan (2), Lisa Ciacchi (2), Hugh H. Reid (2), Jamie Rossjohn (2,3,4§), Lisa A. Chakrabarti (1,5§), Stephanie Gras (2,3§)
(1) Pasteur Institute, Viral Pathogenesis Unit, Paris, France.
(2) Infection and Immunity Program and Department of Biochemistry and Molecular Biology, Biomedicine Discovery Institute, Monash University, Clayton, Victoria 3800, Australia.
(3) ARC Centre of Excellence in Advanced Molecular Imaging, Monash University, Clayton, Victoria 3800, Australia.
(4) Institute of Infection and Immunity, School of Medicine, Cardiff University, Cardiff CF14 4XN, UK.
(5) INSERM, U1108 Paris, France.
*These authors contributed equally to this work as co-first authors.
§These authors contributed equally to this work as co-senior authors.