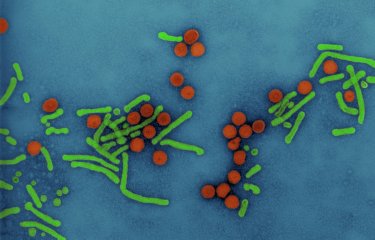
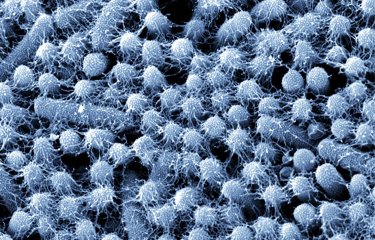
Pathogenic bacteria known as AIEC, which bind to the intestinal mucosa, are frequently found in Crohn's disease patients. Scientists at the Institut Pasteur, in collaboration with scientists from the University of Clermont Auvergne and the Lille Faculty of Pharmaceutical and Biological Sciences, administered a cocktail of bacteriophages orally to murine models for the disease. This led to a reduction in the concentration of AIEC bacteria and to the disappearance of some symptoms, offering new hope for the future treatment of Crohn's disease patients.
Crohn's disease is a chronic inflammation of the digestive tract that is thought to affect approximately one in every thousand people in France. The disease cannot be treated, and it can be difficult to control the associated abdominal pains and other even more debilitating symptoms. Little is known about the causes of this disease. We do know, however, that there are lesions in the digestive tract in the final section of the small intestine, the ileum, located just before the colon. And in approximately 30% of patients, these ileal lesions are colonized by pathogenic bacteria known as AIEC, or adherent-invasive Escherichia coli. These bacteria bind to a receptor (CEACAM6) at the surface of epithelial cells. "We have known for a long time that wherever there are bacteria there are also bacteriophages, including in our intestine, and the role of most of these bacteriophages is to infect and kill bacteria," explains Laurent Debarbieux, Head of the Bacteriophage–Bacteria Interactions in Animals Group at the Institut Pasteur. "In Crohn's disease, the composition of the gut microbiota (the entire population of bacteria in the intestine) is unbalanced. So we are trying to restore this balance by using bacteriophages that will specifically target AIEC bacteria without touching the rest of the microbiota, unlike antibiotics."
This team at the Institut Pasteur studies bacteriophages and their potential therapeutic application in depth. Drawing on their expertise, their goal was to reduce the concentration of AIEC bacteria in the intestinal mucosa in mice. Working with scientists from the University of Clermont Auvergne and the Lille Faculty of Pharmaceutical and Biological Sciences, "we observed that three bacteriophages were able to replicate in the ileum," continues Laurent Debarbieux. "And after just one day of treatment with this cocktail of three bacteriophages, administered orally, we observed a significant reduction in the number of AIEC in feces and within the intestine itself." The scientists also observed that in mice a single administration led to a reduction that lasted for two weeks, together with the alleviation of colitis symptoms.
These findings, confirmed by analyses on human intestinal samples, demonstrate that bacteriophages represent a new treatment option for targeting AIEC bacteria in Crohn's disease patients. This research also provides a strong basis for conducting a clinical trial.
These results were obtained with the support of the DigestScience Foundation and the Ferring International Center SA.
Source
Bacteriophages targeting adherent invasive Escherichia coli strains as a promising new treatment for Crohn’s disease, J Crohns Colitis, January 12, 2017
Matthieu Galtier,1,2* Luisa De Sordi,1* Adeline Sivignon,3 Amélie de Vallée,3 Damien Maura,1,2,4 Christel Neut,5,6 Oumaira Rahmouni,5,6 Kristin Wannerberger, 7 Arlette Darfeuille-Michaud,3 Pierre Desreumaux, 6,8 Nicolas Barnich,3 Laurent Debarbieux1
1: Institut Pasteur, Molecular Biology of the Gene in Extremophiles, Department of Microbiology, F-75015 Paris, France
2: Paris Diderot University, Sorbonne Paris Cité, Pasteur Unit, rue du Docteur Roux, F-75015 Paris, France
3: University of Clermont Auvergne, UMR 1071 Inserm/University of Auvergne, INRA, Unit under contract 2018, Clermont-Ferrand, France
4: Present address: Department of Surgery, Harvard Medical School and Massachusetts General Hospital, Boston, 02114, Massachusetts, United States of America
5: Division of Bacteriology, Faculty of Pharmaceutical and Biological Sciences, University of Lille Nord de France (ComUE), F-59006 Lille Cedex, France
6: Inserm U995 - LIRIC - Lille Inflammation Research International Center, F-59000 Lille, France
7: Ferring International Center SA, Ch. de la Vergognausaz 50, 1162 St-Prex, Switzerland
8: Lille University Hospital, Department of Digestive Tract Diseases and Nutrition, Claude Huriez Hospital, F-59037 Lille, France
*: these authors contributed equally to this work
Mis à jour le 30/01/2017