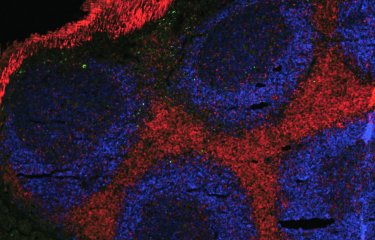
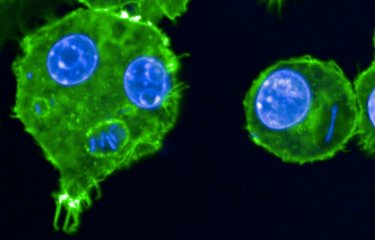
Parasites of the genus Leishmania show important genomic instability, which these pathogens use to adapt to their environment. A study by Institut Pasteur researchers shows that Leishmania exploits non-coding RNAs to mitigate toxic effects of genome instability by post-transcriptional regulation and the establishment of specialized ribosomes.
Darwinian evolution plays a central yet poorly understood role in human disease. Iterations between genetic mutation and environmental selection drive cancer development, microbial infection and therapeutic failures. The molecular mechanisms that harness the deleterious effects of genome instability to generate beneficial phenotypes in pathogens are unknown. In a paper, published in PNAS, scientists from the Molecular Parasitology and Signaling Unit of the Institut Pasteur, with a participation of the Bioinformatics and Biostatistics Hub, and their collaborators have demonstrated that the deleterious effects of genome instability in Leishmania parasites can be compensated by adaptive changes in RNA stability and modification.
The study was conducted in the parasite Leishmania that causes devastating human diseases termed leishmaniases. These infections can reach the viscera and prove fatal. The parasite is transmitted by an insect, the sandfly. Present on all continents, leishmaniasis affects between 700,000 and 1 million people every year. A better understanding of the adaptation mechanisms of Leishmania may represent a way to manage these diseases.
Non-coding RNA affects ribosomes
Leishmania parasites lack transcriptional control and instead regulate the level of gene expression by frequent changes in chromosome and gene copy number, and thus by gene dosage. Analyzing in vitro adaptation of hamster-derived parasites via gene copy number (genomic level) and gene expression (transcriptomic) changes, the teams have linked fitness gain in this unique eukaryotic model system to co-amplification of functionally related genes. A major group of genes that were amplified were shown to code for small nucleolar RNAs (snoRNAs) that are involved in ribosomal RNA (rRNA) processing and modification of cellular RNAs.
Gene dosage-dependent increase in snoRNA abundance was indeed correlated with increased modification of rRNA. Given the role of rRNA to enable protein synthesis, these findings suggest establishment of fitness-adapted ribosomes that may further mitigate the possible toxic effects of genome instability by regulating the translation of messenger RNAs into proteins. The link between Leishmania genome instability, snoRNAs and protein translation proposes non-coding RNAs, like snoRNAs, as potential novel biomarkers with diagnostic and prognostic value that may be linked to changes in parasite tissue preference or its drug susceptibility. This novel insight into Leishmania adaptation will be likely applicable to other fast evolving eukaryotic systems with unstable genomes, such as fungi or cancer cells.
This study is part of the priority scientific area Emerging infectious diseases of the Institut Pasteur's strategic plan for 2019-2023.
Source :
Genome instability drives epistatic adaptation in the human pathogen Leishmania, PNAS, Decembre 21, 2021
Giovanni Bussotti1,2, Laura Piel2, Pascale Pescher2, Malgorzata A. Domagalska3, K. Shanmugha Rajan4, Tirza Doniger4, Disha-Gajanan Hiregange5, Peter J. Myler6-10, Ron Unger4, Shulamit Michaeli4, Gerald F. Späth2
1 - Institut Pasteur, hub de Bioinformatique et biostatistique, C3BI, USR 3756 IP CNRS, Paris, France.
2 - Institut Pasteur, INSERM U1201, unité Parasitologie moléculaire et signalisation, Paris, France.
3 - Institut de médecine tropicale, unité Parasitologie moléculaire, B-2000, Anvers, Belgique.
4 - Faculté des sciences de la vie Mina et Everard Goodman et Institut de nanotechnologie et des matériaux avancés, université Bar-Ilan, Ramat Gan 52900, Israël.
5 - Département de biologie chimique et structurale, Institut Weizmann des Sciences, Rehovot, 7610001 Israël.
6 - Seattle Structural Genomics Center for Infectious Disease, Seattle, Washington, États-Unis.
7 - Department of Biomedical Informatics and Medical Education, université de Washington, Seattle, Washington, États-Unis.
8 - Department of Pediatrics, université de Washington, Seattle, Washington, États-Unis.
9 - Department of Global Health, université de Washington, Seattle, Washington, États-Unis.
10 - Center for Global Infectious Disease Research, Seattle Children's Research Institute, Seattle, Washington, États-Unis.