How understanding the immune system can help us age better

Contents
Report - How understanding the immune system can help us age better
In terms of DNA and immune defenses, we are all different
The "Milieu Intérieur" project: a unique study to elucidate the mysteries of immunity
Definition - What is epigenetics?
Biological sex shapes immunity
Four major findings regarding aging
Revealing how age, sex and vaccination can shape immunity
Definition - Single-cell sequencing
Towards the emergence of personalized aging treatments
Aging is a natural phenomenon that happens to us all – and it goes far beyond merely the number of years we live. With life expectancy on the rise, especially in industrialized countries, the notion of quality of aging – what it means to age well – is increasingly important. By 2050, nearly 10% of the European population will be 80 or over, and the number of centenarians is likely to be 18 times higher than in the year 2000. These developments come with considerable medical and social challenges.
In terms of DNA and immune defenses, we are all different
Although everyone gets older, aging never looks the same from one person to the next. Each individual has a unique immune heritage, passed down from their ancestors, which deeply influences the way in which their body works. The immune system also remembers pathogens that it has protected us from in the past. Its composition, effectiveness and ability to adapt vary considerably from one person to another.
In addition to this genetic diversity, there are also differences related to biological sex. Women and men do not have the same immune defenses or the same aging trajectories. Women generally have a stronger immune response but they are also more exposed to certain autoimmune diseases. Men sometimes experience a steeper immune decline at an earlier age, with specific consequences for long-term health.

Report from the Translational Immunology Laboratory with the LabEx Milieu intérieur team. SiMoA® (Quanterix) is an ultra-sensitive immunoassay technology that enables the detection of proteins and nucleic acids at the lowest possible levels. - Copyright : Thomas LANG 2020 / Institut Pasteur
"Milieu Intérieur" LabEx - A large-scale French research project to understand the variability of the immune response
The "Milieu Intérieur" Laboratory of Excellence (LabEx) was launched for the very purpose of improving our understanding of immune diversity. This vast French research project, involving 45 laboratories, set out to define what a "healthy" immune system is and to measure natural immune variability among a cohort of a thousand healthy adults (see inset on previous page).
In 2021, ten years after the program began, the same cohort was studied again. This longitudinal research offers a rare opportunity to observe how the immune system evolves over time in individuals who were initially in good health, and to elucidate the complex mechanisms that link aging with the immune system.
These mechanisms show us that immune aging is neither linear nor uniform. It is based on the interaction of several biological phenomena that develop at a different pace in each individual.
The Milieu Intérieur project, launched in 2011 at the Institut Pasteur and led by Darragh Duffy and Lluis Quintana-Murci, explores the variability of the immune system in healthy individuals. Unlike research on specific diseases, it monitors a cohort of 1,000 volunteers – 500 women and 500 men, aged between 20 and 70 – over more than a decade through a combination of genetic and immune analysis and environmental data. This approach has revealed predictive markers of disease, immune differences related to sex or age, and the impact of factors like smoking and viral infections.
The results pave the way for more personalized medicine involving tailored treatments (for example for tuberculosis), early detection of metabolic complications and a better understanding of aging mechanisms. The project is now also shedding light on the links between immunity, aging and epigenetics.
By making the data available to the scientific community, the "Milieu Intérieur" project is helping shape the future of health care, in which prevention and precision will play an increasing part.
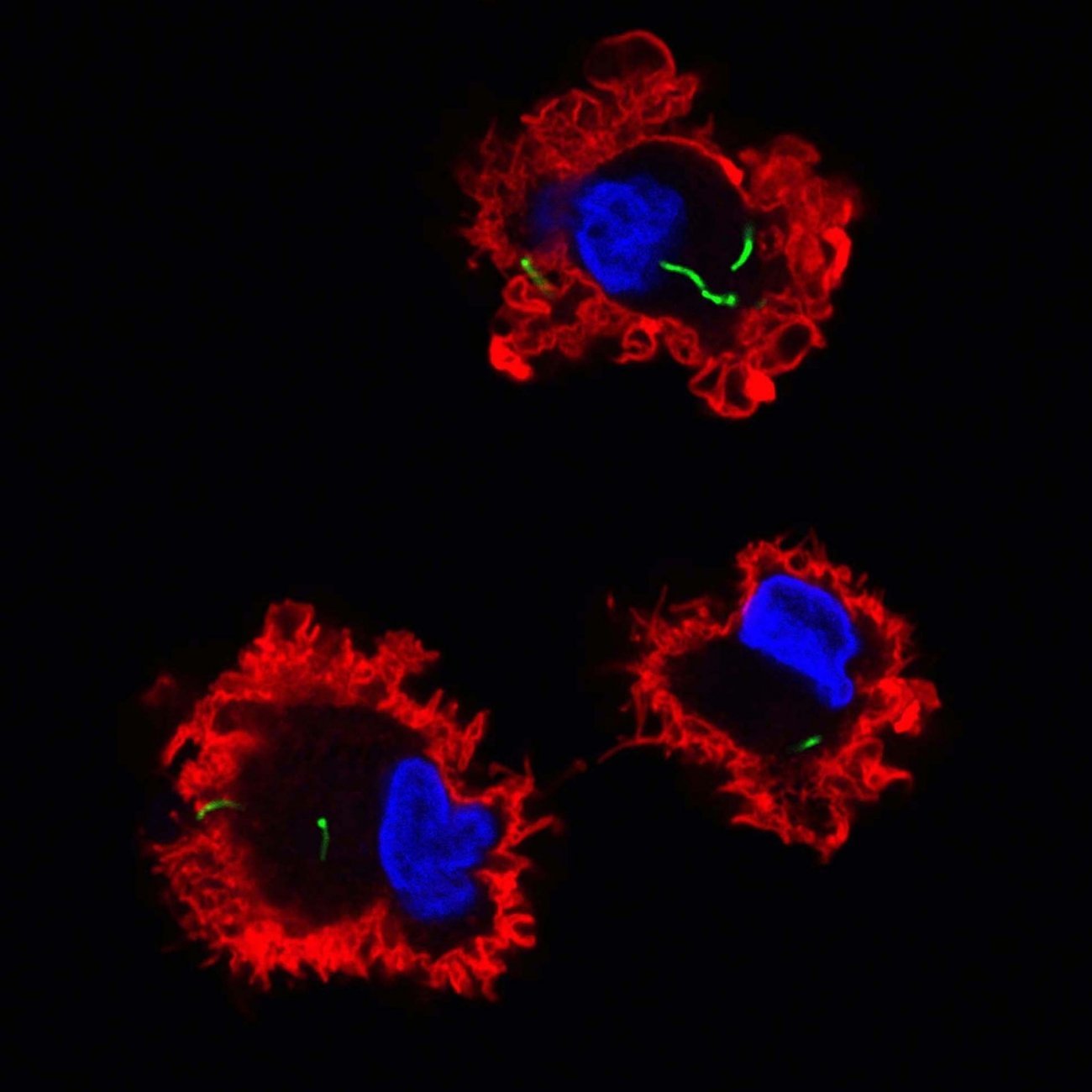
The tuberculosis bacillus (in green) enters dendritic cells (in red) - Copyright: Institut Pasteur/Ludovic Tailleux
What is epigenetics?
Epigenetics is the study of reversible and inherited changes that influence gene activity without changing the sequence of DNA itself. It explores how external factors such as environment, lifestyle, food or stress can activate or deactivate the expression of certain genes, like a light switch. Epigenetic modifications play a crucial role in the body's development and adaptation to its environment.The field of epigenetics is shedding new light on the origins of diseases such as cancer, neurological disorders and diabetes, and paving the way for personalized treatments.
Biological sex shapes immunity
The differences between men and women when it comes to disease are striking. For example, 50% of women will contract a urinary infection during their lifetime, compared with just 10 to 15% of men. Bladder cancer affects three times more men than women, but women with bladder cancer are often diagnosed at a later stage and have a worse prognosis. These disparities raise a basic question: why do immune responses and health trajectories differ depending on sex?
Molly Ingersoll's team is seeking to elucidate this enigma by studying underexplored mucosal sites such as the bladder. The team's theory is based on the influence of steroid hormones (estrogen, progesterone, androgens and cortisol), which act as powerful modulators of the immune system. To test their hypothesis, the scientists worked with the "Milieu Intérieur" consortium and analyzed 17 different hormones in the blood of the cohort volunteers, around 500 of whom came back to provide additional samples ten years later.
INSERER VIDEO MOLLY INGERSOLL
This longitudinal approach offers an insight into how our hormones change as we age. The results are striking.
Female hormonal contraception changes virtually the entire steroid landscape, including cortisol and cortisone, key regulators of stress and immunity. This systemic impact is as yet poorly understood, and it raises questions about long-term effects on metabolism and resistance to infection or other diseases.
The menopause is a critical turning point, with rapidly declining estrogen and progesterone levels associated with increased susceptibility to bladder infections and changes to the characteristics of bladder tumors.
In men who smoke, there was a surprising correlation between nicotine and androgens that was not found in women, revealing a link between specific levels of male hormones and environmental disruptors.
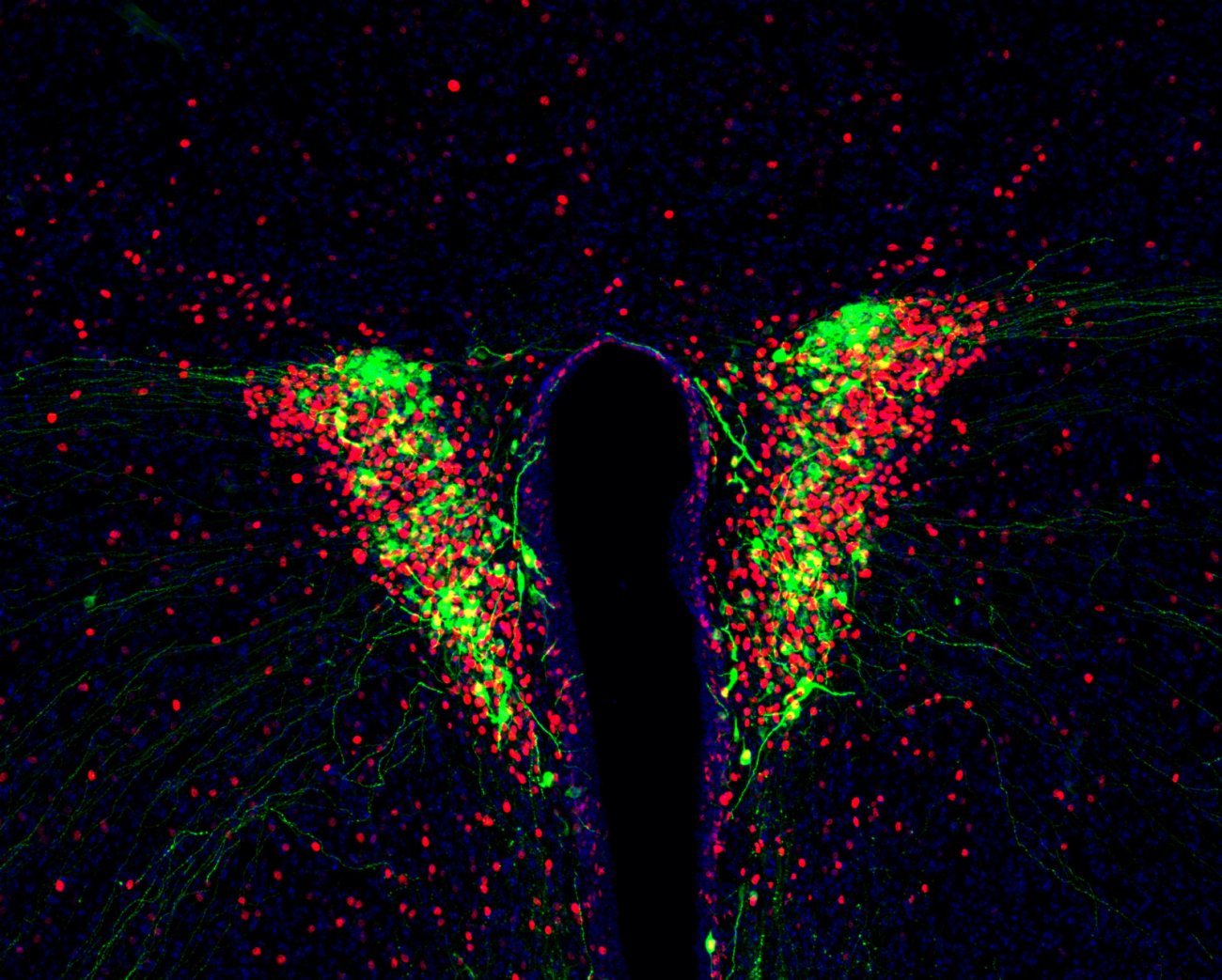
Hormone-producing neurons in the hypothalamus. In the hypothalamus, neurons producing vasopressin (green), a hormone essential for stress response and antidiuretic function, are activated by inflammation in the blood (c-Fos, red). - Copyright: Institut Pasteur/Ferdinand Jagot
Finally, monitoring the cohort over ten years showed that a significant drop in progestogens is unexpectedly linked with a higher risk of disease, but only in men.
The study has its limits – it can reveal correlations but not causality. But the quality of the data sets is unparalleled, providing scientists with rare and valuable insights.
The data will serve as a global benchmark. Future projects will focus on dissociating the effects of hormones from those of X/Y chromosomes and personalizing treatments based on sex. The challenge lies in adapting medicine to the biological reality of women and men, resulting in fairer, more effective therapies for everyone.

DNA acts like a temporal microscope, revealing how human populations have reacted to pathogens over thousands of years.
The "Milieu Intérieur" project is a unique scientific journey. What is your role in the project?
"Milieu Intérieur" is an ambitious project designed to explore the variability of the human immune system by examining immunological, genetic, epigenetic and environmental data. We are seeking to identify the main genetic and environmental factors that affect variations in immune response in the general French population. Within the consortium, my task is to determine the causes of immune aging and to understand how modifications in DNA influence the immune response as we age. I also coordinate the analysis and management of the huge volumes of data generated by the project, making sure that the results are robust and usable.
How is sequencing ancient DNA revolutionizing our understanding of the immune system?
For geneticists, the ability to sequence ancient DNA is as significant as the invention of the microscope for microbiology. We can now directly observe how human populations and their pathogens have evolved over thousands of years, rather than just relying on theoretical models.This approach reveals the causes of past pandemics like the Black Death. When we compared the genomes of victims and survivors buried in Toulouse in the 14th and 15th centuries, it appeared that several genes thought to confer susceptibility to plague were not actually involved. This suggests that non-genetic factors such as nutrition and living conditions played a key role in the pandemic, which wiped out 30 to 60% of the European population.
Was agriculture really the key turning point in the evolution of the human immune system?
Some plausible scientific theories, like the fact that agriculture gave rise to an epidemiological transition, can all too easily become dogmas. Our data show that the Bronze Age was a much more decisive period for the immune system in Europe as it brought urbanization, mass migration and population growth, which were conducive to the spread of pathogens and made natural selection more effective. We need to reconsider these long-held theories to better understand the causes of the demographic crises that have punctuated the history of our species and the evolutionary mechanisms that have shaped our immunity.
You are also working on Polynesian populations. What lessons have you drawn from this research, especially your work on metabolic disorders?
My colleagues Lluis Quintana-Murci, Van-Mai Cao-Lormeau and I sequenced 1,800 modern genomes to study the history of settlement and susceptibility to metabolic disorders. Contrary to what is generally believed, our results do not support the theory that the current obesity epidemic is caused by a higher genetic risk in Pacific populations. It is clearly linked with recent environmental factors, like the sudden shift in dietary patterns.Today's Polynesians eat ultra-processed foods, and their obesity rate has risen from 0% in the 1950s to 50% today. This raises questions about the impact of interactions between genes and environment on modern diseases.
Your research shows that mutations which confer resistance to infections also increase the risk of autoimmune diseases. How do you explain this paradox?
It is not a paradox when we see it through the lens of the theory of evolution. Mutations that once protected us from infections can become harmful in a less infectious environment. For example, a mutation in the TLR1 gene, which boosts resistance to pathogens, is also associated with an increased risk of allergies and asthma. In an ultra-hygienic environment like ours, such mutations no longer confer a selective advantage and can even lead to autoimmune diseases. This phenomenon could be contributing to the frequency of allergies and autoimmune diseases in modern societies.
What are the future challenges for your research?
If we want to gain a better understanding of the history of our health, we need to develop new approaches to identify the causes of death of our ancestors, like estimating age of death based on ancient DNA by using markers associated with aging. We also want to study other epidemics, like those that wiped out up to 90% of Polynesian populations when the Europeans arrived, and also smallpox and Spanish influenza, even if there are technical hurdles (ancient viral genomes are difficult to sequence) and ethical challenges (the descendants of individuals who died recently can be identified). Every ancient skeleton is a piece in the puzzle of our biological history. Our health is the result of a complex history shaped by interactions between our genes and our environment.
Four major findings regarding aging
One key observation is that as we age we gradually develop low-grade chronic inflammation, which has been referred to as "inflammaging." This diffuse inflammation, which is not clinically obvious, occurs with age and can create favorable conditions for the development of various chronic conditions such as cardiovascular disease, type 2 diabetes and some neurodegenerative disorders. It may also contribute to the gradual exhaustion of the immune system, as it constantly calls on the body's defenses.
Another important finding is the impact of aging on autoimmune diseases. Aging can lead to immune dysregulation, meaning that the immune system is less able to tell the difference between the body's own cells and foreign invaders such as viruses or bacteria. The body can then attack healthy cells by mistake, causing unnecessary inflammation. These dysfunctions can result in the emergence or exacerbation of autoimmune disorders, which vary in frequency and intensity from one person to the next, especially depending on sex and heredity.
Persistent viral infections, especially those caused by viruses from the herpesvirus family like cytomegalovirus, are also closely linked with aging. The presence of these viruses, which can remain latent for decades, causes the body to constantly mobilize immune cells to control infection. Over the long term, this continuous immune response depletes the reservoir of immune cells capable of responding to new infectious agents.
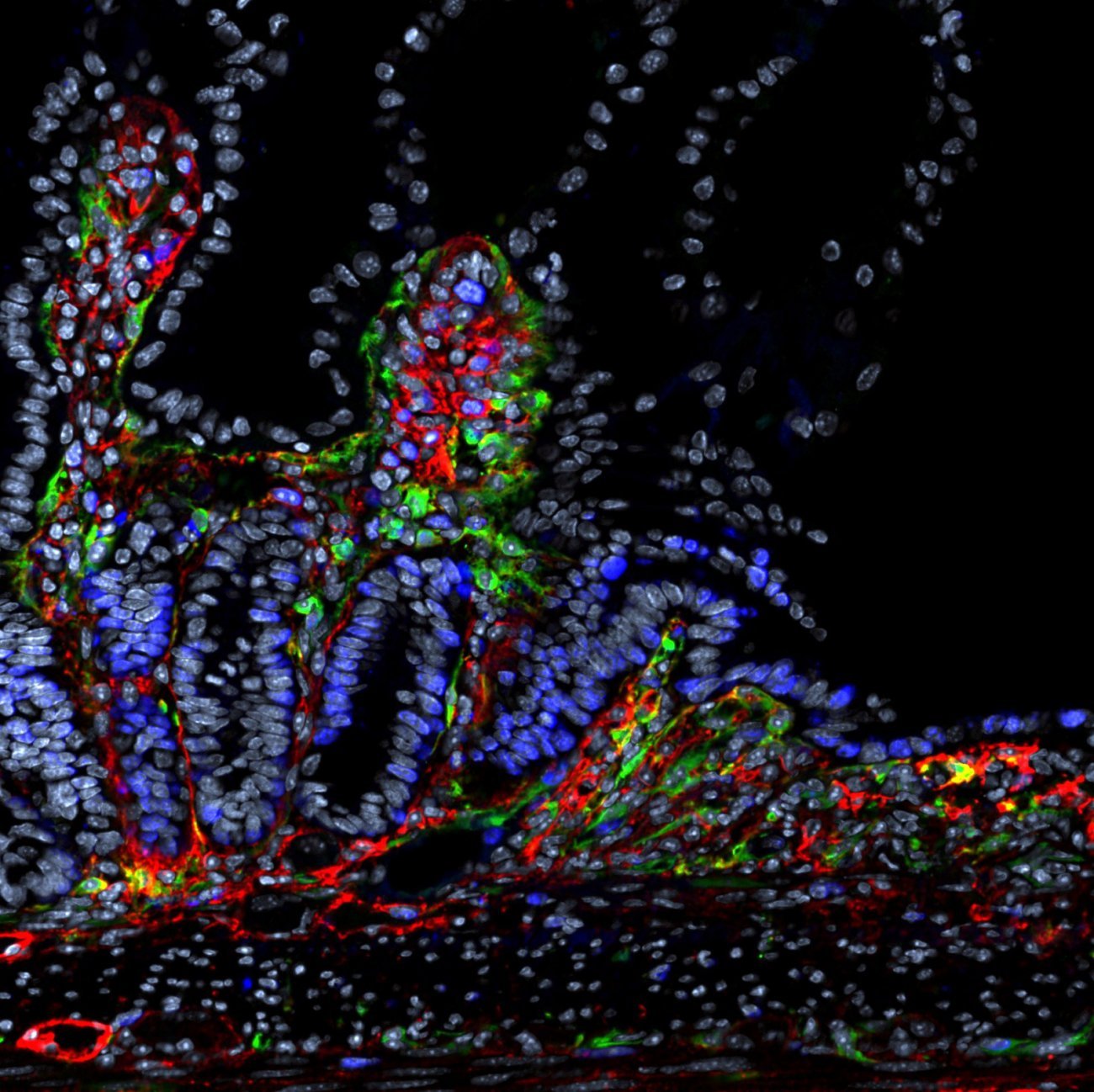
Inflammatory bowel ulcer. Proliferating progenitor cells are shown in blue, stromal cells in red and green, and cell nuclei in gray. Copyright: Institut Pasteur/Lucie Peduto
Lastly, the concept of an epigenetic clock can shed new light on aging by demonstrating that our chronological age does not always reflect the actual state of our cells. Environmental, behavioral or infectious factors can accelerate or slow down biological aging. The epigenetic clock is a promising tool in determining individual trajectories of immune aging and anticipating the risks of age-related conditions. It can be used to estimate the biological age of cells, regardless of their chronological age.
Cellular aging in two people of the same age can look completely different as a result of genetic, environmental and behavioral factors. Using the epigenetic clock concept can be an effective means of predicting the development of the immune system and identifying potential areas for action.
Revealing how age, sex and vaccination can shape immunity
Single-cell sequencing
Single-cell sequencing is a state-of-the-art technique that analyzes RNA cell by cell, revealing cell diversity and individual cell function. It uses microfluidic systems to identify cell subpopulations such as treatment-resistant tumor cells or rare subpopulations that emerge as we age and cannot be detected with conventional methods. In oncology, it can be used to distinguish between healthy and malignant cells and to pinpoint therapeutic targets. This approach is revolutionizing diagnostics, personalized treatment and our understanding of disease.
By exposing the blood cells of 380 individuals in the "Milieu Intérieur" cohort to pathogens like SARS-CoV-2 or the influenza virus in vitro, the scientists demonstrated that elderly people had a weaker immune response, in particular as a result of lower production of interferon alpha, a protein that plays a crucial role in blocking viral replication. "Genomic analysis also showed that CD4 T cells are the cells most affected by aging," continues the scientist.
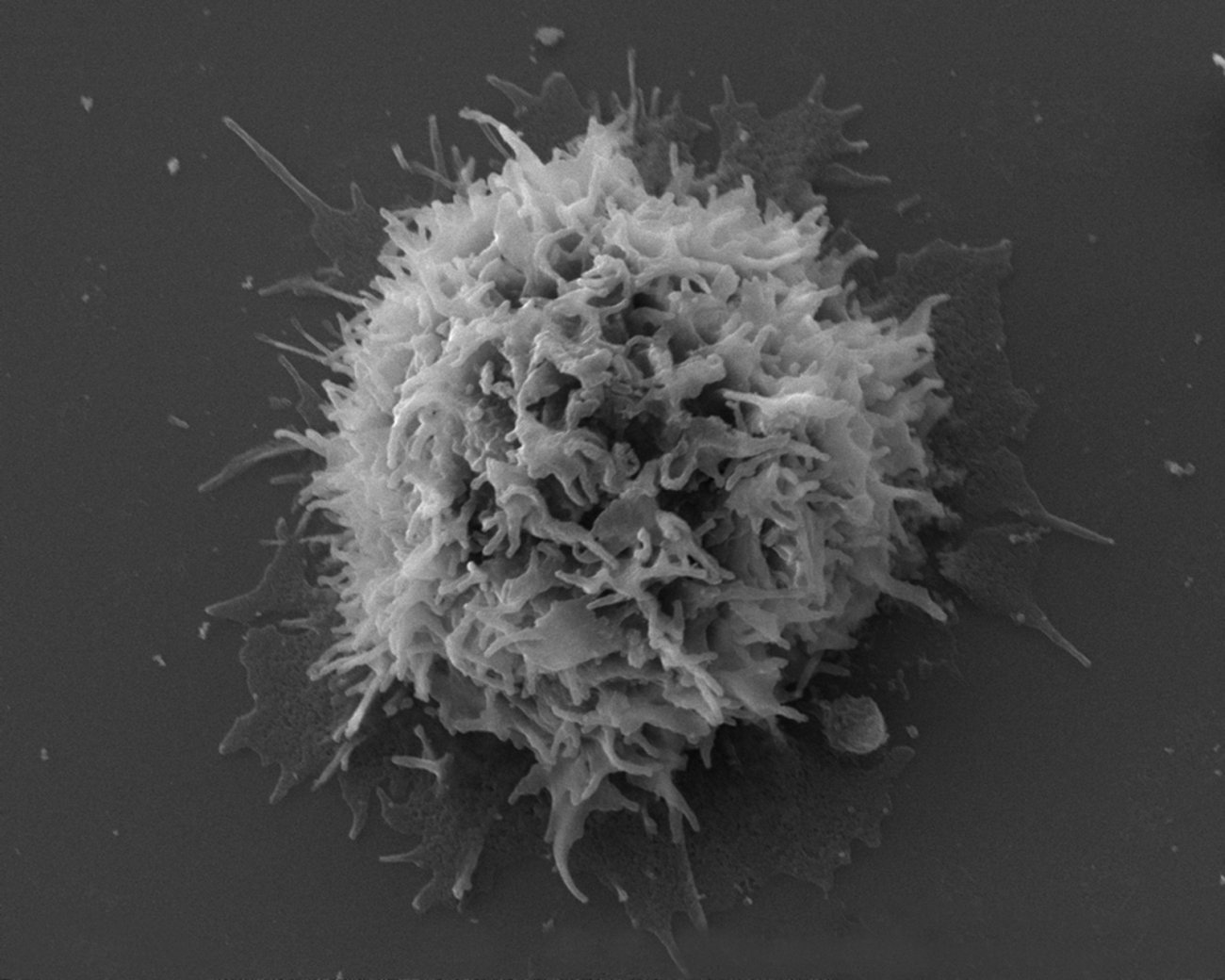
CD4 T lymphocyte viewed under a scanning electron microscope. - Credit: Institut Pasteur/Olivier Schwartz and Electron Microscope Platform.
The research also confirms that women have a stronger immune response than men. This phenomenon is known as the morbidity-mortality paradox: although women live longer on average, they are also more likely to develop autoimmune diseases. "In women, innate immune cells like myeloid cells react more strongly to viruses, with a higher expression of proinflammatory genes," explains the scientist. This can partly be explained by their two X chromosomes. Some genes escape X-chromosome inactivation – the mechanism whereby one of the two X chromosomes in females is shut off –, and this gives them an immunological advantage.
Conversely, men over the age of 60 often lose their Y chromosome (up to 50% in some cases), a phenomenon that the team linked to a weaker immune response for the first time. "The loss of the Y chromosome could explain why men become more vulnerable to severe infections as they get older," says Marwan Sharawy. These discoveries could pave the way for us to maintain a strong immune system even as we age. "If we can understand why some older people still have an effective immune system, we might be able to reproduce those conditions in others," suggests Marwan Sharawy. These results could also inspire novel therapeutic approaches, like targeted stimulation of interferon alpha. "The aim is not just to live longer, but to reach old age in better health," he sums up.

Report on the Biomics technology platform - Copyright: Alexandre Darmon / Art in Research
Towards the emergence of personalized aging treatments
All these observations lead us to a clear conclusion: with humans displaying such a variety of immune profiles and aging trajectories, we can no longer rely on a standardized approach to health. Understanding aging means taking into account the biological age, genetic heritage, infectious history and lifestyle of each individual.
This will pave the way for more personalized medicine based on the identification of precise biomarkers, which will be used to assess the state of the immune system, detect imbalances at an early stage and develop tailored prevention and treatment strategies. Clinicians will be able to propose bespoke vaccination schedules, target persistent viral infections more effectively, and introduce specific measures to reduce chronic inflammation.
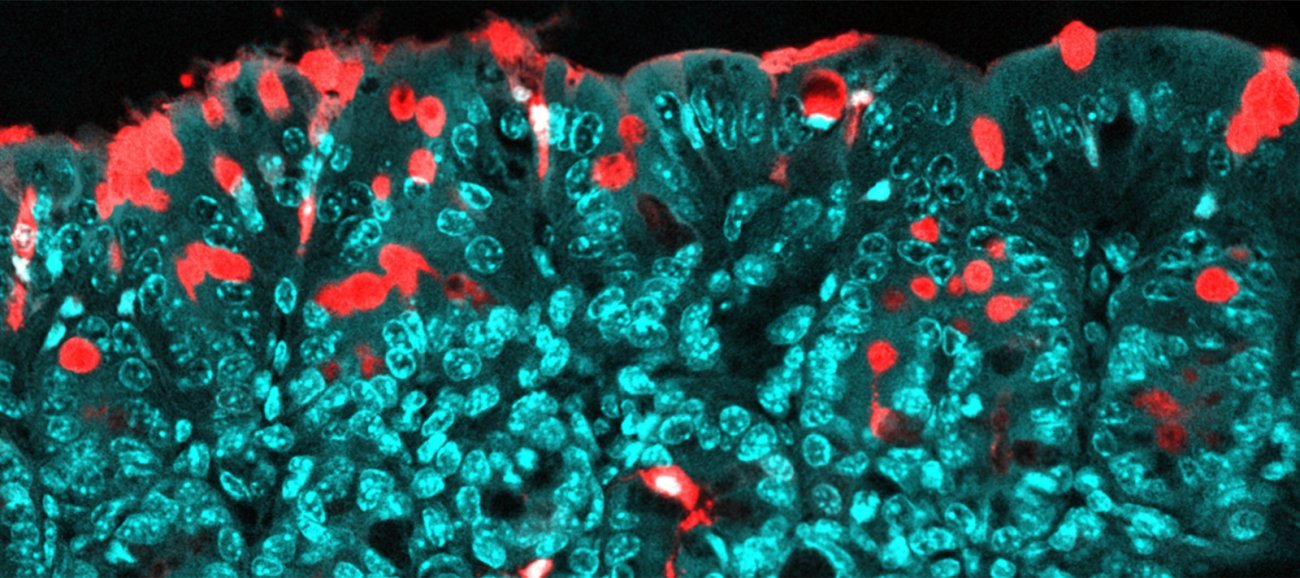
Passage of molecules through the colon epithelium via specialized pathways (goblet cells). In red: transported molecules (antigens); in blue: the epithelium. - Copyright: Institut Pasteur/Microbiota-Host Interactions/Clara Delaroque and Benoit Chassaing
As well as prevention, personalized medicine will also be used to optimize treatments based on the specific characteristics of each patient. The aim is twofold: to improve the effectiveness of medical treatment and reduce unwanted side effects, while also helping each and every one of us to stay more healthy as we get older. Given the wide range of immune profiles and aging trajectories, medicine needs to become more personalized and offer tailored solutions, such as precision immunization, personalized nutritional guidelines or targeted lifestyle adjustments. Potential innovations also include promising therapeutic avenues like gene or epigenetic therapy, which could ultimately "rewind" our biological clock. By targeting chronic inflammation or stimulating immune cell regeneration, these approaches could help us live longer, healthier lives, far beyond just adding years to our age.
Smoking leaves a lasting mark on the immune system
Smoking has a lasting impact on the immune system, even up to 40 years after quitting. Nicotine influences our immune defenses to the same extent as age or sex, by modifying the production of cytokines, key proteins in the immune response.
The long-term impact of smoking is a result of epigenetic modifications (alterations in gene expression that do not change DNA) affecting immunomodulators. Even after years of abstinence, the immune profile of former smokers can still resemble that of active smokers for some immune responses. The message is clear: it is never too late to stop smoking, but it is even better not to start in the first place.
Analysis of the "Milieu Intérieur" cohort also revealed two other factors that have a major impact on our immune system. The first is human cytomegalovirus (HCMV), a very common virus that confers lasting changes on some immune cells. The second is excess weight: a high body mass index (BMI) disrupts the production of key immune messengers like interleukin-2. To isolate these effects, the scientists stimulated blood cells with pathogens and linked the results with 136 variables including sociodemographic and lifestyle factors. Advanced statistical analysis confirmed that smoking, HCMV and BMI influence immunity regardless of age and sex, with persistent DNA damage in former smokers.

Report from the Translational Immunology Laboratory with the LabEx Milieu Intérieur team. Violaine Saint-André presents the bioinformatics work carried out to analyze the Milieu Intérieur dataset. - Copyright: Thomas LANG 2020 / Institut Pasteur
These findings could have major implications for public health. The team is now studying the influence on immunity of the exposome – everything that we are exposed to in our lives, such as pollution, diet, stress, chemicals and viruses –, and trying to identify the metabolic and epigenetic pathways involved. "Understanding how the environment shapes immunity can provide us with more targeted prevention strategies," says Violaine Saint-André. " Our gene expression is highly regulated," concludes the scientist.
Our choices and our environment matter, and identifying what we can do, like quitting smoking and eating healthily, can make a meaningful difference when it comes to preserving our health.
Living longer, healthier lives
Future approaches to treating aging will involve a combination of prevention, personalization and targeted therapies. Anti-inflammatory or antiviral treatments that are currently under development, together with a better understanding of our DNA, could revolutionize quality of life for older people. The aim is not just to live longer but to age in good health, with a resilient immune system and a body that is better equipped to respond to age-related diseases.