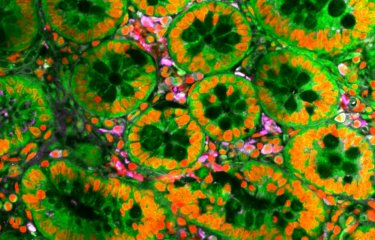
Leishmaniasis is a human and animal disease caused by parasites of the Leishmania genus that affects 12 million people worldwide. It is endemic in around a hundred countries and is currently emerging in Europe as a result of climate change and large-scale population movements. Scientists from the Institut Pasteur in Paris, in collaboration with the Institut Pasteur of Shanghai-Chinese Academy of Sciences, have elucidated a new resistance mechanism of the infectious agents of leishmaniasis against host antimicrobial activities. The discovery paves the way for the development of new therapies to eliminate the harmful influence of the parasite.
Leishmaniasis is one of the world's five leading parasitic diseases. Some 350 million people are thought to be exposed to the risk of infection. The clinical signs of the disease vary – from disfiguring cutaneous forms to fatal visceral forms – depending on which parasite species is responsible for the infection (see the fact sheet in French). Given the global incidence of leishmaniasis, it is one of the world's most neglected diseases. There is no human vaccine and treatment options are limited. "Improving our understanding of human infection with Leishmania is vital if we are to tackle this public health challenge," explains Gérald Spaeth, Head of the Molecular Parasitology and Signaling Unit at the Institut Pasteur in Paris.
Macrophages, key cells in Leishmania infection
In their hosts (rodents, dogs and humans), the parasite causing leishmaniasis develop and proliferate in immune cells, mainly macrophages. Macrophages are part of the innate immune system (the body's first line of defense against infection) and are often exploited as host cells by viral and bacterial pathogens. The pathogens then develop strategies to modulate the macrophage phenotype to ensure their own survival.
Scientists from the "Inflammation and Leishmania Infection" Pasteur International Joint Research Unit (PIU), which links the Institut Pasteur in Paris and the Institut Pasteur of Shanghai, Chinese Academy of Sciences, set out to understand the mechanisms of interaction between Leishmania and macrophages to elucidate how these pathogens resist macrophage antimicrobial activity.
Epigenetic modifications behind Leishmania survival
Using animal models, the scientists analyzed the ability of macrophages to activate a protein complex known as the inflammasome, which triggers inflammatory processes to combat infection.
"We observed that Leishmania is capable of inhibiting inflammasome activation and therefore countering the immune response," explains Gérald Spaeth. The scientists also successfully identified the mechanisms responsible for this inhibition. The mechanism relies on what is known as a dichotomous regulation: it reduces the expression of the positive regulators that activate the inflammasome, while also activating the expression of the inflammasome's negative regulators. "The most remarkable finding is that the modulations to this macrophage function are epigenetic in origin (reversible changes in gene expression)," notes Gérald Spaeth.
By manipulating gene expression, Leishmania is able to survive in the host macrophage without activating its inflammasome – a remarkable strategy, since the parasite persists in the macrophages on a long-term basis and enables these cells to cope with a substantial parasite load. "It is the first time that such an epigenetic strategy has been described for this parasite, since most infectious agents activate inflammasomes," observes Guangxun Meng, Head of the Innate Immunity Unit at the Institut Pasteur of Shanghai, Chinese Academy of Sciences.
Mimicking the suppressive effect of the parasite on the inflammatory response could serve as a basis for the development of new therapeutic options for the treatment of chronic diseases, some forms of cancer and autoimmune disorders. "These findings improve our knowledge on the control of the NF-kB signaling pathway," confirms Dr. Robert Weil, from the Center for Immunology and Infectious Diseases (CIMI) in Paris.
The results of this research open up promising new avenues for the development of anti-Leishmania drugs that target the host epigenome rather than the parasite itself.
---------------------------------------------------
Source
Targeting Macrophage Histone H3 Modification as a Leishmania Strategy to Dampen the NF-kB/NLRP3- Mediated Inflammatory Response,Cell Reports, February 11, 2020
Hervé Lecoeur1,2,3,8, Eric Prina1,3,8, Thibault Rosazza1,3, Kossiwa Kokou1,2,3, Paya N’Diaye1, Nathalie Aulner4, Hugo Varet5, Giovanni Bussotti5, Yue Xing2,3, Geneviève Milon6, Robert Weil7, Guangxun Meng2,3, Gerald F. Späth1,3,9
1 INSERM U1201, Molecular Parasitology and Signaling Unit, Department of Parasites and Insect Vectors, Institut Pasteur, 25 Rue du Dr Roux, 75015 Paris, France
2 Institut Pasteur of Shanghai, The Center for Microbes, Development and Health, Key Laboratory of Molecular Virology & Immunology, Chinese Academy of Sciences, Shanghai 200031, China
3 Institut Pasteur International Mixed Unit Inflammation and Leishmania infection, Paris, France
4 Photonic BioImaging Technology and Service Unit, Center for Technological Resources and Research, Department of Technology and Scientific Programs, Institut Pasteur, 28 Rue du Dr Roux, 75015 Paris, France
5 Bioinformatics and Biostatistics Hub, Department of Computational Biology, Institut Pasteur, USR 3756 CNRS, Paris, France
6 Institut Pasteur, 25 Rue du Dr Roux, 75015 Paris, France
7 Sorbonne University Association, French National Institute for Health and Medical Research (INSERM, UMR1135), French National Center for Scientific Research (CNRS, ERL8255), Center for Immunology and Infectious Diseases CIMI, Paris, France