Cells from the rare individuals who naturally control HIV infection have been the focus of investigation for nearly 15 years with the aim of elucidating their specific features. Following research on the ANRS CO21 CODEX and CO6 PRIMO cohorts, scientists from the Institut Pasteur have described the characteristics of CD8 immune cells in these "HIV controller" subjects. The unique antiviral power of these immune cells can be attributed to an optimal metabolic program that confers persistence and the ability to react effectively against infected cells. Working ex vivo, the scientists successfully reprogrammed cells from infected non-controller individuals to give them the same antiviral potency as controllers' cells. Their findings are published in the journal Nature Metabolism on July 12, 2019.
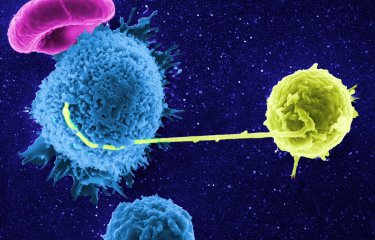
Some people have the ability to control HIV naturally, without treatment. In these very rare individuals (less than 1% of people living with HIV), no multiplication of the virus in the blood can be detected after more than 10 years of infection without treatment. In 2007, scientists from the Institut Pasteur described the extraordinary antiviral activity of these patients' CD8 lymphocytes. Unlike in non-controllers, the CD8 cells of HIV controllers are able to rapidly destroy infected CD4 cells.
Scientists from the Institut Pasteur's HIV, Inflammation and Persistence Unit continued their research with the aim of identifying the specific characteristics of these cells so that they could confer the same characteristics on the cells of non-controller subjects.
The CD8 cells (or "memory cells") of controllers outwardly appear to be identical to those of non-controllers. But the scientists demonstrated that they have a different molecular program. Their research shows that anti-HIV CD8 cells in controllers not only have huge antiviral potential; they are also programmed to survive, whereas in non-controllers, the cell program predisposes them to exhaustion and cell death.
The CD8 cells of controllers use a variety of metabolic resources, drawing in particular on the energy supplied by their mitochondria,[1] which enables the cells to survive in conditions of stress. Conversely, the cells of non-controllers depend on a single energy source (glucose) and they have limited mitochondrial activity. "We identified that the antiviral activity of CD8 cells in controllers is associated with an optimal program that gives them plasticity in using the cell's energy resources," explains Asier Saez-Cirion, a scientist in the Institut Pasteur's HIV, Inflammation and Persistence Unit and coordinator of the study.
In the laboratory, the scientists then managed to stimulate mitochondrial activity in the anti-HIV cells of non-controllers. They used a substance secreted by the immune system known as interleukin 15 (IL-15) to boost the mitochondrial activity of non-controllers' cells and increase their anti-HIV potential. The reprogrammed CD8 cells of non-controllers are able to destroy infected CD4 cells, much like controllers' cells.
"Our research shows that even if the anti-HIV CD8 cells of non-controllers are relatively ineffective compared with those of controllers, the differences can be overcome," concludes Asier Saez-Cirion.
Metabolic reprogramming of immune cells is a strategy that is already being tested in clinical trials for cancer treatment. The scientists hope to be able to test the anti-HIV capabilities of the strategy in vivo in the near future.
This research was funded by the Institut Pasteur and by the ANRS, MSDAVENIR, the European Union (Horizon 2020) and Sidaction.
1Mitochondria: organelles inside cells that provide the energy needed for cells to function properly.
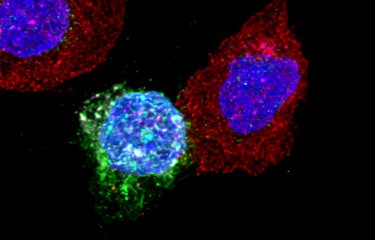
Caption : CD8 lymphocytes (red) of HIV controller patients in contact with CD4 cells (green) infected with HIV. The cell nuclei are in blue. In pink, a cytotoxic molecule secreted by CD8 cells to destroy CD4 cells.
Source
Metabolic plasticity of HIV-specific CD8+ T-cells is associated with enhanced antiviral potential and natural control of HIV-1 infection, Nature Metabolism, 12 juillet 2019
Mathieu Angin1, Stevenn Volant2, Caroline Passaes1, Camille Lecuroux3, Valérie Monceaux1, Marie-Agnes Dillies2, José Carlos Valle Casuso1, Gianfranco Pancino1, Bruno Vaslin3, Roger Le Grand3, Laurence Weiss4,5, Cecile Goujard6, Laurence Meyer7, Faroudy Boufassa7, Michaela Müller-Trutwin1, Olivier Lambotte3,6, and Asier Sáez-Cirión1
1 Institut Pasteur, Unité HIV Inflammation et Persistance, Paris, France
2 Institut Pasteur, Hub Bioinformatique et Biostatistique – C3BI, USR 3756 IP CNRS – Paris, France
3 CEA, Université Paris Sud, INSERM U1184, Immunology of Viral Infections and Autoimmune Diseases (IMVA), IDMIT Department / IBFJ, 92265 Fontenay-aux-Roses, France
4 Assistance Publique Hôpitaux de Paris, Hôpital Européen Georges Pompidou, Paris, France
5 Université Paris Descartes, Sorbonne Paris Cité, Paris, France
6 Assistance Publique Hôpitaux de Paris, Hôpital Bicêtre, Service de Médecine Interne et Immunologie clinique, 94275 Le Kremlin-Bicêtre, France
7 INSERM U1018, Centre de recherche en Epidémiologie et Santé des Populations, Université Paris Sud, Le Kremlin Bicêtre, France