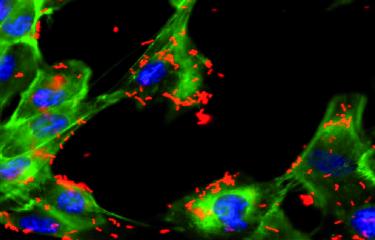
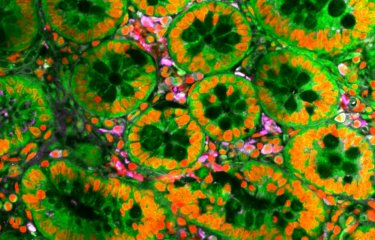
Scientists from the Institut Pasteur have contributed to the discovery of a mechanism that regulates the response to pneumococcal infections between asymptomatic and severe forms. Pneumococci are bacteria that are naturally present in the human respiratory tract, but they are also one of the main causes of meningitis, pneumonia and acute otitis media.
According to Santé publique France,1 the mortality rate following a pneumococcal infection is 10 to 30%, and more than 800,000 children worldwide die from pneumococcal infections each year. Although Streptococcus pneumoniae bacteria, or pneumococci, can cause very severe respiratory infections, they also reside in many people asymptomatically, without causing any apparent symptoms.
Some pneumococcus strains, known as carrier strains, give rise to asymptomatic forms and are mainly found in the upper respiratory tract. Others, known as invasive strains, are more frequently responsible for more severe infections, in which the bacteria spread in the lungs, the blood or the meninges.
Scientists do not yet know exactly what regulates the transition between asymptomatic and invasive forms.
Discovery of the role of the KDM6B protein
Scientists conducted a comparative study of the responses induced by a carrier strain and an invasive strain. Mélanie Hamon, Head of the Chromatin and Infection Unit at the Institut Pasteur, who led the study, explains: "We revealed a novel process used by pneumococci to maintain the lifestyle of an asymptomatic colonizer. The carrier strain does this by increasing expression of a protein known as KDM6B in the invaded host cells." The scientists also observed that KDM6B plays a role in the regulation of a gene (IL-11) during infection with the carrier strain.
An epigenetic regulation mechanism
The KDM6B protein is an enzyme that modifies eukaryotic DNA compaction. It was already known for its role in cell development and the regulation of inflammatory responses. The scientists found that KDM6B is key to the regulation of the asymptomatic or inflammatory response during pneumococcal infection.
When the activity of KDM6B was reduced experimentally, the cells infected by the carrier strain, which would normally express an asymptomatic form of infection, became symptomatic, as if they had been infected by the invasive strain. The opposite phenomenon occurred when the activity of KDM6B was mimicked by adding IL-11 to a cell infected by the invasive strain. "The bacteria use what is known as an 'epigenetic' mechanism to actively maintain the integrity of the infected cells," explains Mélanie Hamon.
This research represents a breakthrough in our understanding of non-pathogenic bacteria-host interactions, which could have a broader impact on the development of new therapies to tackle respiratory infections.
Sources :
(1) Les infections à pneumocoque sur Santé publique France.
(2) The histone demethylase KDM6B fine-tunes the host response to Streptococcus pneumoniae, Nature Microbiology, 21 December 2020
Michael G. Connor 1, Tiphaine Marie-Noelle Camarasa1,2, Emma Patey1, Orhan Rasid 1, Laura Barrio 3,4, Caroline M. Weight 5, Daniel P. Miller6, Robert S. Heyderman 5, Richard J. Lamont 7, Jost Enninga3,4 and Melanie A. Hamon 1
1G5 Chromatin and Infection, Institut Pasteur, Paris, France.
2Université Paris Diderot, Sorbonne Paris Cité, Paris, France.
3Dynamics of Host-Pathogen Interactions Unit, Institut Pasteur, Paris, France.
4UMR CNRS, Paris, France.
5Division of Infection and Immunity, University College London, London, UK.
6Department of Microbiology and Immunology, Virginia Commonwealth University School of Medicine, Richmond, Virginia, USA.
7Department of Oral Immunology and Infectious Diseases, School of Dentistry, University of Louisville, Louisville, Kentucky, USA.
.