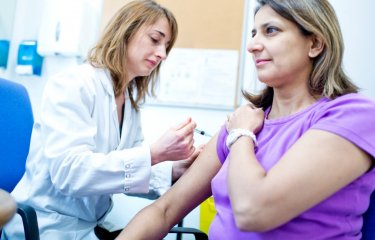
The results of a clinical trial aimed to assess the effectiveness of an oral vaccine against Shigella dysenteriae serotype 1, the bacterium responsible for the epidemic form of Shigellosis or bacillary dysentery, a severe and often fatal diarrheic disease, have just been published in the journal Vaccine. Developed by a team from the Institut Pasteur and Inserm, this vaccine proved to be well tolerated and to elicit a specific immune response that may confer protection against the most severe symptoms of Shigellosis after a single oral administration. The Phase II trial was carried out by the Institut Pasteur in cooperation with Inserm and the Paris Network of Public Hospitals (AP-HP). The researchers have henceforth the hope develop a vaccine effective and easy to administer against the epidemic fits of bacillary dysentery occuring in humanitarian dramas (disasters, civil wars, refugee camps), mostly due to this serotype that is resistant to most of the antibiotics.
Press releease
Paris, january 15, 2009
Shigellosis, or bacillary dysentery, caused by several serotypes of the bacterium Shigella, is responsible every year for hundreds of thousands of deaths in the developing countries, particularly at the young children, according to the World Health Organization. This severe invasive intestinal infection is characterized by bloody diarrhea. Shigella dysenteriae serotype 1 is responsible for the most virulent form of the disease, causing brutal and uncontrollable epidemics marked by very high rates of attack and mortality with regard to the other serotypes of Shigella and to the other digestive pathogens in general. The multidrug resistance of this serotype to antibiotics, including fluoroquinolones, leaves without weapon against this disease in the absence of an effective vaccine .
Several years of research in the Molecular Microbial Pathogenesis Unit, led by Philippe Sansonetti at the Institut Pasteur (Inserm 786), allowed the development of a candidate vaccine against Shigella dysenteriae type 1. This live attenuated vaccine was obtained by targeted inactivation of certain genes that contribute to the virulence of the bacterium. “Immunity can be acquired naturally following shigellosis, as has been observed in endemic areas,” explains Philippe Sansonetti. “Given that our candidate vaccine is able to mimic the natural process by which protective immunity is triggered, we hoped that it would be effective.”
This vaccine has been given the name “SC599”. After a Phase I trial which allowed to test its non-toxicity, the Institut Pasteur launched in 2006 a Phase II trial to assess its immunogenicity. 111 volunteers have been recruited in two vaccinology centers: the Cochin-Pasteur Vaccinology Center (CIC) at Cochin Hospital, Paris, coordinated by Dr. Odile Launay, and St. George’s Vaccine Institute in London, led by Dr. David Lewis. It was a randomized double-blind placebo-controlled trial.
The volunteers were divided into three groups. One received a placebo. The other two groups were given a dose of the candidate vaccine at a different concentration. For about 40% of the vaccinees, both doses proved to be immunogenic, capable of triggering the production of cells producing specific antibodies against the bacterium.
“The immune response generated in these volunteers suggests that SC599 vaccine might offer some protection for people exposed to the disease in endemic areas,” explains Marie-Lise Gougeon, one of those in charge of the trial and the leader of the Antiviral Immunity, Biotherapy and Vaccines Unit at the Institut Pasteur. “The protective power of this vaccine can only be really estimated by subsequent field trials.”
The next step is the launch of a second Phase II trial, already in preparation. It will aim at using two vaccinal doses and not a unique dose. The booster dose will be administered some weeks after the initial dose. “The boost should in theory, increase the number of subjects who respond to the vaccine,” explains Marie-Lise Gougeon.
The objective of the researchers is to manage to make of the candidate SC599 an effective vaccine for most large number, little expensive and easy to use on the ground, because administered by oral route.
This trial was able to be carried out thanks to funding from the Institut Pasteur and the Direction Générale de l’Armement.
Sources:
“Safety and immunogenicity of SC599, an oral live attenuated Shigella dysenteriae type-1 vaccine in healthy volunteers: results of a phase 2, randomized, double-blind placebo controlled trial”: Vaccine, 2009, in press.
Odile Launay 1, Christine Sadorge 2, Nathalie Jolly 2, Béatrice Poirier 3, Stéphane Béchet 2, Diane van der Vliet 1, Valérie Seffer 3, Nicola Fenner 4, Kelly Dowling 4, Raphaela Giemza 4,
Julie Johnson 4, Anna Ndiaye 2, Muriel Vray 5, Philippe Sansonetti 6, Philippe Morand 7, Claire Poyart 7, David Lewis 4, Marie-Lise Gougeon 3
1- University Paris-Descartes; Inserm; Cochin-Pasteur Vaccinology CIC (CIC BT505); Pôle de Médecine; Cochin-Saint Vincent de Paul Hospital Group, Paris, France
2- Vaccine and Biomedical Research Center, Institut Pasteur, Paris, France
3- Antiviral Immunity, Biotherapy and Vaccines Unit, Institut Pasteur, Paris, France
4- St George’s University of London, London SW17 ORE, UK
5- Epidemiology of Emerging Diseases Unit, Institut Pasteur, Paris, France
6- Molecular Microbial Pathogenesis Unit, Institut Pasteur, Inserm Unit 786, Paris, France
7- Bacteriology Department, Cochin-Saint Vincent de Paul Hospital Group; Paris Public Hospital Network (AP-HP); National Reference Center for Streptococci, Paris, France.
Contact persons:
- Institut Pasteur Press Office : Corinne Jamma or Nadine Peyrolo –
+33 (0)1 40 61 33 41 – cjamma@pasteur.fr
- Inserm Press Office: Séverine Ciancia – +33 (0)1 44 23 60 86 – presse@inserm.fr
- AP-HP Communication Division – Press Office: +33 (0)1 40 27 37 22 – service.presse@sap.aphp.fr