A new endogenous viral element (EVE) has been identified in Aedes aegypti mosquitoes. This EVE is almost identical to a contemporaneous strain of cell-fusing agent virus (CFAV), a widespread insect-specific virus found in Ae. aegypti populations. Researchers experimentally demonstrated that this EVE controls CFAV replication in the mosquito’s ovaries. This discovery supports the idea that EVEs constitute a universal system of heritable sequence-specific antiviral immunity.
Endogenous viral elements (EVEs), also known as viral fossils, are viral sequences integrated in host genomes. When viral DNA integration occurs in the germline, it can be inherited and retained in the host genome as evidence of ancient viral infections.
Retrovirus-derived EVEs are the best-known examples as they play important roles in host physiology and antiviral immunity. Recent bioinformatic surveys also identified non-retroviral EVEs in a wide range of animal genomes including mosquitoes.
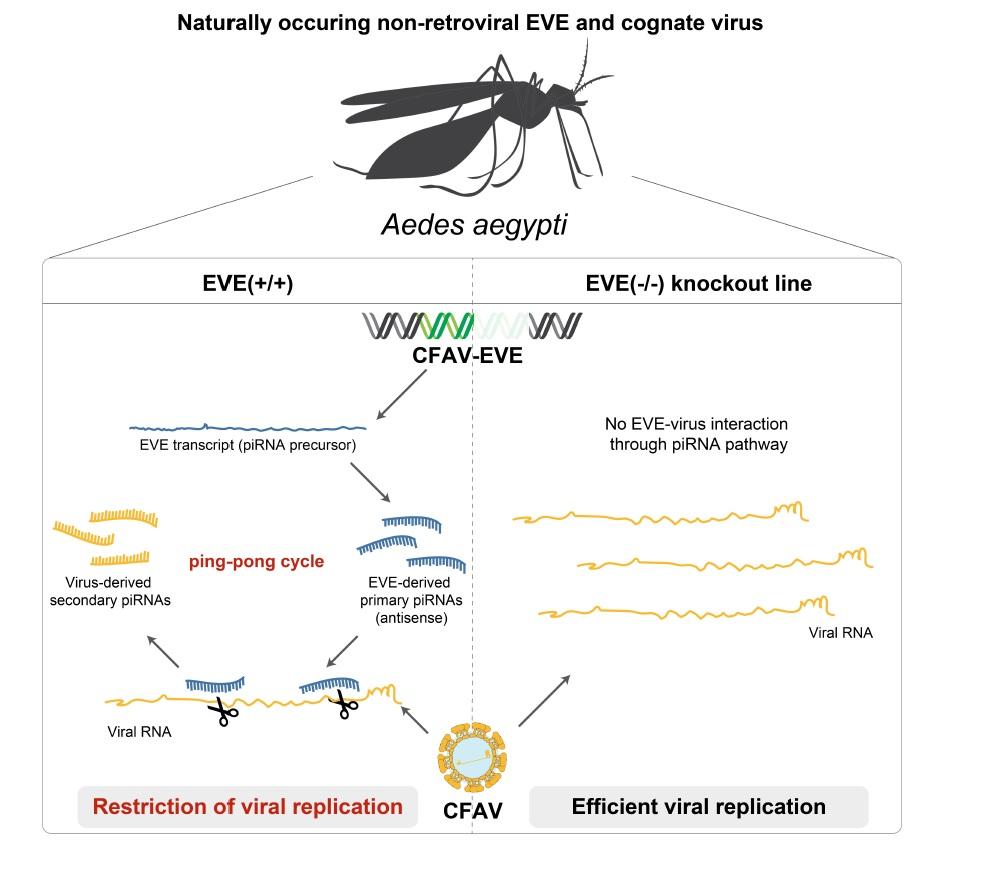
Non-retroviral EVEs produce piRNAs, a class of small RNAs canonically associated with genome integrity through the control of transposons in the germline. This observation has led to the hypothesis that non-retroviral EVEs could also play a role in the antiviral immunity of eukaryotes.
A major challenge to prove this hypothesis is that the viruses currently circulating generally do not share a high nucleotide identity with the corresponding EVE sequences, preventing a possible match between EVE-derived piRNAs and the target viral RNA.
“We identified a new EVE in Aedes aegypti mosquitoes that is 96% identical at the nucleotide level to a contemporaneous strain of cell-fusing agent virus (CFAV), a widespread insect-specific virus in Ae. aegypti populations,” explains Louis Lambrechts, Head of the Insect-Virus Interactions Unit at the Institut Pasteur (Paris).
Using this naturally occurring insect-virus interaction, the researchers showed that the EVE and the viral RNA interact through the piRNA pathway. Ablation of the EVE by CRISPR/Cas9 genome editing resulted in increased CFAV replication in mosquito ovaries.
According to Carla Saleh, Head of the Viruses and RNA Interference Unit at the Institut Pasteur (Paris), “our results reveal the antiviral activity of a non-retroviral EVE against its cognate virus. We propose that EVEs constitute a universal system of heritable, sequence-specific antiviral immunity in eukaryotes, analogous to CRISPR/Cas immunity in prokaryotes.”
Source
Non-retroviral endogenous viral element limits cognate virus replication in Aedes aegypti ovaries, Current Biology, July 16, 2020
Yasutsugu Suzuki1*, Artem Baidaliuk2,3*, Pascal Miesen1,4, Lionel Frangeul1, Anna B. Crist2, Sarah H. Merkling2, Albin Fontaine2, Sebastian Lequime5, Isabelle Moltini-Conclois2, Hervé Blanc1, Ronald P. van Rij4, Louis Lambrechts2**, Maria-Carla Saleh1**
1. Viruses and RNA Interference Unit, Institut Pasteur, UMR3569, CNRS, Paris, France
2. Insect-Virus Interactions Unit, Institut Pasteur, UMR2000, CNRS, Paris, France
3. Sorbonne Université, Collège doctoral, F-75005 Paris, France
4. Department of Medical Microbiology, Radboud Institute for Molecular Life Sciences, Radboud University Medical Center, Nijmegen, The Netherlands
5. KU Leuven Department of Microbiology and Immunology, Rega Institute, Laboratory of Clinical and Epidemiological Virology, Leuven, Belgium
*These authors contributed equally.
**These authors contributed equally.
This study is part of the Vaccinology and Immunotherapy Initiative of the Institut Pasteur's strategic plan for 2019-2023.