We now know that neurodegenerative diseases are caused by the aggregation of "misfolded" proteins in the brain. These proteins, which are toxic for neurons, spread from cell to cell through tiny tunnels, known as nanotubes. After imaging their structure in detail, (1) scientists in the Institut Pasteur's Membrane Traffic and Pathogenesis Unit, directed by Chiara Zurzolo, are now investigating the mechanisms by which these nanotubes are formed and the propagation of abnormal alpha-synuclein proteins associated with Parkinson's disease. The results of their research have been published in the journals European Molecular Biology Organization and Neurobiology of Disease.
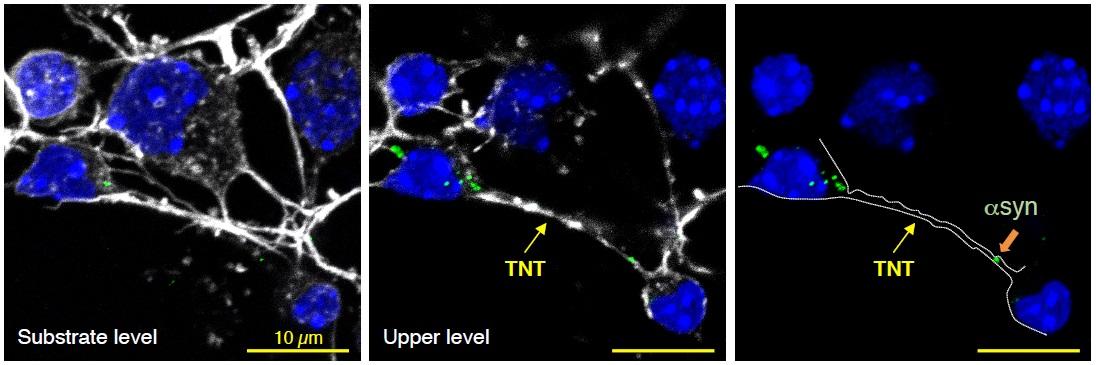
In the brain, neurons communicate with each other by means of billions of synaptic connections. But they can also exchange information via what are known as tunneling nanotubes (TNTs). These tiny tunnels – tentacles ranging from 20 to 500 nanometers in diameter and up to 100μm long – enable neurons and other cells in the body to exchange signaling molecules, proteins and organelles such as mitochondria – but also pathogens. Scientists in the Membrane Traffic and Pathogenesis Unit, directed by Chiara Zurzolo at the Institut Pasteur, discovered that the amyloid proteins that cause neurodegenerative diseases use TNTs to circulate between cells, ultimately destroying them. In Parkinson's disease, the aggregated alpha-synuclein protein uses this network of nanotubes to spread and accumulate within neurons. "We hypothesized that this is a key mechanism used to spread the pathology to the entire brain, and that it therefore plays a vital role in the progression of Parkinson's disease," explains Chiara Zurzolo. “Therefore, it is crucial for us to understand how TNTs are formed so that we can develop therapeutic strategies to curb the progression of the disease."
In a new study, Chiara Zurzolo's team therefore decided to investigate the mechanisms responsible for TNT formation and regulation in neurons. The scientists used cultures of mouse primary neurons to identify the molecular players involved in the growth of these actin-based cell extensions. The first finding was that the Wnt/Ca2+ signaling pathway, vital for cell development, also plays a key role in the formation of nanotubes and their stability in neurons. Using super-resolution microscopy, which enables visualization of nanoscopic structures, the scientists revealed that CAM-kinase II proteins (2) bind to the actin cytoskeleton at the base of nanotubes, thereby contributing to the stability of these structures and increasing their life span. Another finding was that activation of the Wnt/Ca2+ pathway enhances the transfer of alpha-synuclein aggregates between neurons, and this transfer is disrupted in mutant mice. The presence of "misfolded" alpha-synuclein aggregates appears to trigger the formation of tunnels that are useful for the proteins' own dissemination in the brain. "This study backs up our theory that TNT connections are present in the early stages of development, and they can also form later on in adults, in mature brains, if triggered by harmful stimuli such as the presence of amyloid proteins," explains Chiara Zurzolo.
In a second study, the scientists attempted to elucidate what happens when stem cells are transplanted into Parkinson's disease patients, as therapeutic approach. When induced pluripotent stem cells (3) are transplanted, only half of them will differentiate and give rise to dopaminergic neurons that can fill the gaps in the diseased brain and improve symptoms. Previous studies had already demonstrated that abnormal alpha-synuclein proteins in the host brain were capable of "contaminating" grafts. This time, Chiara Zurzolo's team took the investigations a step further and examined neural precursor cells derived from induced pluripotent stem cells in vitro. The scientists firstly confirmed that alpha-synuclein aggregates could enter human neural cells. They then demonstrated that these cells could destroy the toxic proteins – much like astrocytes, glial cells that are particularly effective in destroying pathogenic agents –, but could also transfer them to other cells and neurons via nanotubes. In conclusion, neural precursor cells, in which the presence of nanotubes was revealed for the first time, can play a dual role in the progression of the disease.
This research opens up new avenues for the development of treatments to block the spread of alpha-synuclein, by targeting the Wnt/Ca2+ signaling pathway, for example, or by optimizing the outcome of transplants in patients with Parkinson's disease.
(1) When the structure of tunneling nanotubes (TNTs) challenges the very concept of cell
(2) Proteins regulated by the Ca2+/calmodulin complex
(3) Pluripotent stem cells obtained in the laboratory from somatic cells taken from the patient
Sources
The Wnt/Ca2+pathway is involved in interneuronal communication mediated by tunneling nanotubes, The EMBO Journal, October 18, 2019
Jessica Y Vargas1,†, Frida Loria1,†, Yuan-Ju Wu1,†, Gonzalo Córdova2, Takashi Nonaka3,Se bastien Bellow4, Sylvie Syan1, Masato Hasegawa3, Geeske M van Woerden5,6, Capucine Trollet2 & Chiara Zurzolo1*
1. Unité de Trafic Membranaire et Pathogénèse, Département de Biologie Cellulaire et de l’Infection, Institut Pasteur, Paris, France
2. Institut National de la Santé et de la Recherche Médicale, Association Institut de Myologie, Centre de Recherche en Myologie, UMRS974, Sorbonne Université, Paris, France
3. Department of Dementia and Higher Brain Function, Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan
4. BioAxial, Paris, France
5. Department of Neuroscience, Erasmus Medical Center, Rotterdam, The Netherlands
6. ENCORE Expertise Center for Neurodevelopmental Disorders, Erasmus Medical Center, Rotterdam, The Netherlands
†These authors contributed equally to this work
*Corresponding author
Human NPCs can degrade α–syn fibrils and transfer them preferentially in a cell contact-dependent manner possibly through TNT-like structures, Neurobiology of Disease, September 5, 2019
Clara Grudina1, Georgia Kouroupi2, Takashi Nonaka3, Masato Hasegawa3, Rebecca Matsas2, Chiara Zurzolo1*
1. Unité de Traffic Membranaire et Pathogénèse, Institut Pasteur, 28 Rue du Dr. Roux, Paris 75015, France
2. Laboratory of Cell and Molecular Neurobiology–Stem Cells, Department of Neurobiology, Hellenic Pasteur Institute, 127 Vassilissis Sofias Avenue, Athens 11521, Greece
3. Dementia Research Project, Tokyo Metropolitan Institute of Medical Science, 2-1-6 Kamikitazawa, Setagaya-ku, Tokyo 156-8585, Japan
*Corresponding author
This study is part of the priority scientific area Brain connectivity and neurodegenerative diseases of the Institut Pasteur's strategic plan for 2019-2023.