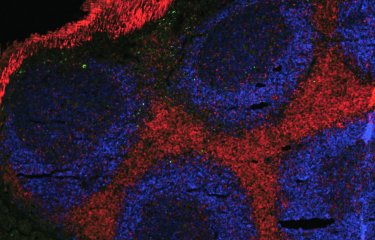
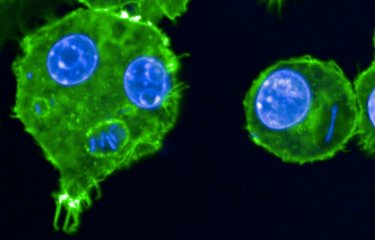
A stem cell model allows researchers to observe the earliest stages of sex determination in humans. This could help uncover why some people are born without a clearly identifiable sex at birth, as well as the development of future fertility treatments.
Very little is known about what happens when an individual’s genetic makeup doesn’t match their physical sex. This is manifested as a spectrum of conditions termed differences of sex development (DSDs), which occur in 1 in 4,500 newborns and is often associated with infertility. Gonad development in human is poorly understood since the genes involved are not conserved in evolution and the tissue from early developing gonad is difficult to access in the human. Progress in this field has been incremental due to the absence of an appropriate, accessible model system.
Researchers at the Institut Pasteur (Anu Bashamboo in the Human Developmental Genetics Unit, led by Kenneth McElreavey), the Francis Crick Institute (Robin Lovell-Badge) and Bar-Ilan University (Nitzan Gonen), have developed a model of early gonad development, using induced pluripotent stem cells, which can be directed to become any cell type in the body. From these cells, using new culture protocols, they generated somatic gonadal cells that go on to become the precursors of either ovaries or testes. Using their model, the researchers at Pasteur grew somatic gonadal cells with male chromosomes (XY), carrying a genetic variant of a specific gene known to cause sex reversal. These cells were unable to form 3D testicular tissue on specially designed microfluidic device, and appeared closer to ovarian cells.
In recent years the Pasteur team has identified several new genes involved in human sex-determination (e.g. Genetic cause of difference in sexual development uncovered). However, they lacked a suitable biological model to study the mechanisms by which the variants in these genes could cause human pathologies of the testis. Combined with genome editing techniques, the new model now allows researchers to study the roles of a range of different genes thought to be involved in sex determination, gonad development and associated pathologies including DSD and infertility.
Their discovery could not only help uncover why some people are born without a clearly identifiable sex at birth, but this new model could be used as a platform for spermatogenesis leading to the development of future fertility treatments.
Read more on Stem cell model allows researchers to observe the earliest stages of sex determination in humans | Crick
Source :
In vitro cellular reprogramming to model gonad development and its disorders, Science advances, January 4, 2023.
DOI: 10.1126/sciadv.abn9793
Nitzan Gonen1,2†*, Caroline Eozenou3, †, Richard Mitter4, Maëva Elzaiat3, , Isabelle Stévant1,
Rona Aviram1, Andreia Sofia Bernardo2,5, Almira Chervova6, Somboon Wankanit3, Emmanuel Frachon7, Pierre-Henri Commère8, Sylvie Brailly-Tabard9, Léo Valon10, Laura Barrio Cano8, Romain Levayer10, Inas Mazen11, Samy Gobaa7, James C. Smith2, Kenneth McElreavey3*, Robin Lovell-Badge2*, Anu Bashamboo3*
1. The Mina and Everard Goodman Faculty of Life Sciences and the Institute of Nanotechnology and Advanced Materials, Bar-Ilan University, Ramat Gan 5290002, Israel.
2. The Francis Crick Institute, 1 Midland Road, London NW1 1AT, UK.
3. Institut Pasteur, Université de Paris, CNRS UMR3738, Human Developmental Genetics, F-75015 Paris, France.
4. Bioinformatics Core, The Francis Crick Institute, 1 Midland Road, London NW1 1AT, UK.
5. National Heart and Lung Institute, Imperial College London, London, UK.
6. Department of Stem Cell and Developmental Biology, Institut Pasteur, Paris 75724, France.
7. Biomaterials and Microfluidics Core Facility, Institut Pasteur, F-75015 Paris, France.
8. Cytometry and Biomarkers, Centre de Ressources et Recherches Technologiques (C2RT), Institut Pasteur, F75015 Paris, France.
9. Assistance Publique-Hôpitaux de Paris, Bicêtre Hospital, Molecular Genetics, Pharmacogenetics, and Hormonology, Le Kremlin-Bicêtre, France.
10. Institut Pasteur, Université de Paris, CNRS UMR3738, Cell Death and Epithelial Homeostasis, F-75015 Paris, France.
11. Genetics Department, National Research Center, Cairo, Egypt.
†These authors contributed equally to this work.