Nicolas Reyes' team has successfully stabilized and determined the first nearly-atomic resolution structure of a human excitatory neurotransmitter transporter, a key component of the synaptic machinery in the brain. This historic study provides hope for potential pharmacological solutions that could selectively modulate these transporters and thereby control the concentration of glutamate – one of the molecules that they transport. Acting on glutamate concentrations in this way could provide an opportunity for treating several neurodegenerative diseases.
Members of the glutamate transporter family, or SLC1, are transmembrane proteins that move excitatory neurotransmitters in the brain, and also amino acids in the peripheral organs. The 3D structure of a model prokaryotic transporter has been known since 2004, but "nobody until now has been able to observe a human SLC1 protein, because they are too unstable under laboratory conditions," explained Nicolas Reyes, who heads up the Molecular Mechanisms of Membrane Transport laboratory*, and whose team just successfully stabilized the human protein.
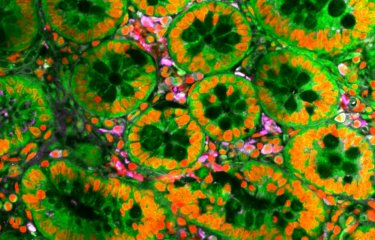
Crystal structure of thermostabilized human EAAT1 showing the Scaffold Domain (green mesh) and the Transport Domain (orange surface) that moves the neurotransmitter glutamate across the membrane in an "elevator-like" fashion”. © Nico Reyes / Institut Pasteur
An SLC1 family with implications for health
"Glutamate is the main excitatory neurotransmitter in the brain, but it is also highly cytotoxic when its extracellular concentration is increased, a pathological mechanism associated with several neurodegenerative diseases”, continued Nicolas Reyes. "Glutamate transporters pump the transmitter into the cells and are key regulators of its extracellular concentration, hence the transporters are important pharmaceutical targets to reduce the concentration of glutamate under pathological conditions.” A high concentration of extracellular glutamate is observed in patients with Alzheimer's disease, although the nature of this correlation is not known. However, acting on the concentration of glutamate may be an avenue for patient treatment.
In addition, in the peripheral organs, glutamine is a key metabolite for transforming healthy cells into malignant cells: here, too, a transporter from the SLC1 family helps maintain normal levels of intracellular glutamine, which is used as a nutrient for the cells. However, in several forms of human cancer, including breast and lung, the SLC1 glutamine transporter is produced in larger amounts to provide the cancer cells with the necessary “dietary“ requirements for their explosive growth.
"It would be really powerful to be able to act pharmacologically on glutamine transport for cancer therapy, and our work opens the possibility for rational drug design of glutamine transport inhibitors", says Nicolas Reyes. A deep understanding of the human SLC1 transporters function is therefore essential, but it cannot be achieved without observing their 3D structure.
Unprecedented atomic precision
Nicolas Reyes' team has determined the first 3D structures of a human SLC1 transporter, answering a question that has eluded researchers since the cloning of neurotransmitter transporters in the 90’s. "We can now view these important human proteins at nearly atomic resolution," stressed Nicolas Reyes. "Our structures will help us to understand the mechanisms of this family of transporters, and their regulation by lipids like cholesterol." Pharmacological solutions (allosteric modulators) can now be envisaged for specifically inhibiting or stimulating SLC1 transporters and modulating the concentration of excitatory neurotransmitters and important amino acids in the human body. Thereby, it would be possible to treat certain neurodegenerative diseases or cancers.
3D structure of Excitatory Amino Acid Transporter 1. © Nico Reyes / Institut Pasteur
This work serves as a reminder of the importance of fundamental research in the study of mechanisms in living beings. " Function without Structure is a ghost, and Structure without function is a corpse” Nicolas Reyes said rephrasing Professor Stephen Wainwright (Duke University, USA). “Our atomic-scale observations provide the basis to understanding real protein function, but we continuously challenge our structural results with functional assays." Why are some SLC1 transporters exclusively selective for glutamate and others for glutamine? What are the mechanisms that inhibit or potentiate their transport function? These are some of the questions that will be addressed in forthcoming research.
* This laboratory is a “Five year group” – a research team comprising young scientists with very high potential.
Source
Structure and allosteric inhibition mechanism of excitatory amino acid transporter 1, Nature, April 19, 2017.
Juan C. Canul-Tec1,4♯, Reda Assal1,4♯, Erica Cirri1,4, Pierre Legrand2, Sébastien Brier3,4, Julia Chamot-Rooke3,4 & Nicolas Reyes1,4
1. Molecular Mechanisms of Membrane Transport Laboratory, Institut Pasteur, 25–28 rue du Docteur Roux, 75015 Paris, France
2. Synchrotron SOLEIL, L'Orme des Merisiers, 91192 Gif-sur-Yvette, France
3. Structural Mass Spectrometry and Proteomics Unit, Institut Pasteur, 25–28 rue du Docteur Roux, 75015 Paris, France
4. UMR 3528, CNRS, Institut Pasteur, 25–28 rue du Docteur Roux, 75015 Paris, France
♯These authors contributed equally to this work