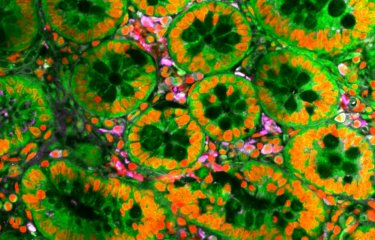
A publication in eLife from researchers of the Institut Pasteur, CNRS, University of Lille and Indiana University (USA) explains the mechanisms by which the bacterium Chlamydia trachomatis hijacks nutrients of its host to its own benefit.
Chlamydia trachomatis is the most common bacterial cause of sexually transmitted infection. These infections are often asymptomatic and are a major cause for infertility. Chlamydia trachomatis is also able to colonize the eye, and the resulting inflammation is the leading cause of blindness by an infectious agent in several developing countries.
Many parasites do no live free in a host cell, but remain inside an internal compartment closed by a membrane, called a vacuole. The vacuole protects the parasites from some host defense mechanisms. However, it blocks direct access to nutrients, and the parasites must develop complex mechanisms to acquire those. In the case of Chlamydia trachomatis, it is known that the vacuole is rich in glycogen, an energy storage molecule made of sugar units.
In this work, researchers from the Institut Pasteur, CNRS, University of Lille and Indiana University explain the mechanisms by which glycogen accumulates around the bacteria. They demonstrate that the glycogen in the vacuole has two origins. Firstly, the bacterium hijacks the glycogen stores synthesized by the host, which get trapped inside the vacuole. Secondly, it synthesizes glycogen directly in the vacuole, using sugar units obtained from the host. As a result, the whole glycogen stores of the infected cell become trapped within the vacuole. This dual strategy fulfills the nutritional needs of the bacteria while weakening the host, which loses access to its own energy stores.
« This discovery answers a 50-year old question: how do the bacteria manage to accumulate glycogen in a space that is separated from the host? We still need to understand the advantage that glycogen accumulation brings to Chlamydia trachomatis, a pathogen that develop exclusively in humans. Indeed, Chlamydia species that infect other hosts do not accumulate glycogen. Understanding the underlying reasons could help us develop novel strategies to fight Chlamydia trachomatis infections » says Agathe Subtil, director of the Unit Cell biology of microbial infection at the Institut Pasteur.
Source
Sequestration of host metabolism by an intracellular pathogen, eLife, march 16, 2016
Lena Gehre (1,2), Olivier Gorgette (3), Stéphanie Perrinet (1,2), Marie-Christine Prévost (3), Mathieu Ducatez (4), Amanda M. Giebel (5), David E. Nelson (6), Steven G. Ball (4), and Agathe Subtil (1,2)
(1) Institut Pasteur, Unité de Biologie cellulaire de l’infection microbienne, 25 rue du Dr Roux, 75015 Paris, France
(2) CNRS UMR3691, Paris, France
(3) Institut Pasteur, Plate-forme de Microscopie Ultrastructurale, Imagopole, Paris, France
(4) Université de Lille, CNRS UMR 8576 - UGSF- Unité de Glycobiologie Structurale et Fonctionnelle, F 59000 Lille, France
(5) Department of Biology, Indiana University, Bloomington, Indiana, USA
(6) Department of Microbiology and Immunology, Indiana University School of Medecine, Indianapolis, Indiana, USA
Mis à jour le 25/03/2016