Scientists from CNRS, the Institut Pasteur (France), the Vlaams Instituut voor Biotechnologie and the Vrije Universiteit Brussel (Belgium) have published a ground-breaking study of the structure and function of a central protein in the liver: NTCP, a cellular-entry pathway for bile salts, but also for certain hepatitis viruses. Published in the journal Nature, these results reveal the 3D structure of NTCP and two architectutres it can adopt. One may be helpful in the development of therapeutic tools against hepatitis viral infection..
Though an essential gateway to the liver, NTCP had not been well described until now. Na+-taurocholate co-transporting polypeptide (NTCP) is a protein located exclusively in the membrane of liver cells that enables recycling of bile acid molecules. It is also the cellular receptor of human hepatitis B and D viruses (HBV/HDV). A better understanding of NTCP could enable the development of treatments specifically designed for the liver, and to fight HBV and HDV infection.
NTCP is a difficult protein to study. It weighs only 38 kilodaltons (kDa)1, whereas cryo-electron microscopy, the technology used to study this type of molecule, only works for molecules weighing more than 50 kDA. The challenge was therefore to “enlarge” and stabilise it.
To do this, teams from French and Belgian laboratories2developed and tested a collection of antibody fragments targeting NTCP. The 3D structures of the resulting complexes were determined using cryo-electron microscopy, and different antibody fragments stabilised and revealed several forms of NTCP.
The research team was able to describe two essential NTCP conformations: one in which the protein opens a large membrane pore to bile salts, to which HBV and HDV can bind, and a second, ‘closed’ conformation, that prevents recognition by the viruses.
The first, ‘open’ conformation is very surprising, as no other known molecular transporter forms such a ‘wide open’ pore. In turn, the second conformation could help finding antiviral molecules that prevent HBV and HDV infection. The research team intends to continue its work to fully elucidate the functioning of NTCP.
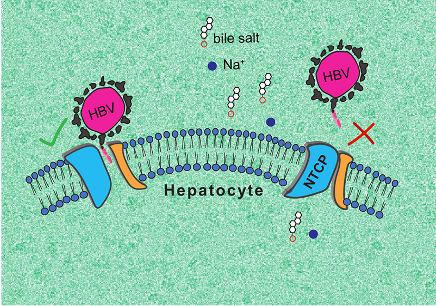
Illustration of the two 3D conformations adopted by NTCPs.
Left: ‘open’ conformation to which HBV and HDV can bind. Right: ‘closed’ conformation that prevents recognition by the viruses.
© Kapil Goutam/Nicolas Reyes/CNRS
Source
Structural basis of sodium-dependent bile salt uptake into the liver.
Kapil Goutam1,2, Francesco S. Ielasi2, Els Pardon3,4, Jan Steyaert3,4 et Nicolas Reyes1,2. Nature, 11th May 2022.
1Membrane Protein Mechanisms Group, European Institute of Chemistry and Biology, University of Bordeaux, CNRS-UMR5234, 33607 Pessac, France.
2Membrane Protein Mechanisms Unit, Institut Pasteur, 75015 Paris, France
3Structural Biology Brussels, Vrije Universiteit Brussel, VUB, Brussels, Belgium 4VIB-VUB Center for Structural Biology, VIB, Brussels, Belgium
1- One dalton is one-twelfth the mass of a carbon-12 atom (the mass of a hydrogen atom, approximately).
2- The study was conducted by teams at the MPF Laboratory (Microbiologie fondamentale et pathogénicité) (CNRS/University of Bordeaux), the Membrane Protein Mechanisms Unit at the Institut Pasteur, and the VIB-VUB Center for Structural Biology. This study was supported by the ANRS Emerging Infectious Diseases Program, among others.


