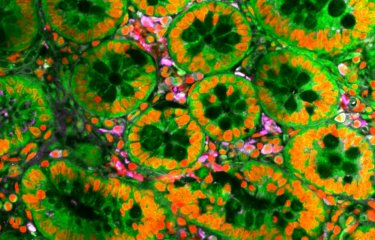
Legionella pneumophila is an opportunistic human pathogen that is increasingly recognized as an important cause of both community and nosocomially acquired pneumonia (Legionnaires’ disease or legionellosis) worldwide. Researchers from the Institut Pasteur tried to understand how a harmless environmental bacterium can become a feared human pathogen.
This human disease represents up to 5% of community-acquired pneumonia with a high mortality rate (11% to 33%), in particular for immunocompromised and elderly people. Risk factors for developing Legionnaires’ disease are smoking, chronic heart or lung disease, diabetes, organ transplantation, immunosuppression, cancer, and age >50 years. Many of these risk factors are increasing in developed countries, as did the number of cases of Legionellosis in the last years (data from ECDC). Outbreaks affecting hundreds of people are frequently reported.
Astonishing evolutionary trajectories
L. pneumophila belongs to a genus that comprises over 60 species, all of which are environmental bacteria that replicate in aquatic protozoa (a unicellular organism) in sweet water ponds and rivers. To understand how a harmless environmental bacterium can become a feared human pathogen, researchers from the Biology of intracellular bacteria Unit at the Institut Pasteur used functional and comparative genomics to deconstruct an entire bacterial genus and reveal for the first time the surprising parallel evolutionary trajectories that have led to the emergence of the major human disease agent, Legionella pneumophila.
Their research has uncovered an unexpectedly large repository of effector proteins (that are proteins secreted in the host cell where they perform and action) with evidence that Legionella species have acquired proteins from all domains of life (plant, animal, fungal, archaea) and have integrated them in their bacterial genomes. The bacteria have transformed these proteins, using them as “tools of oppression” to hijack host cellular functions, in particular targeting signal transduction, protein turnover (balance between protein synthesis and protein degradation) and chromatin modifying pathways.
A trail to explore
“This is the first demonstration of such a massive scale of horizontal gene transfer taking place between protozoa and their bacterial parasites, leading to the emergence of new human pathogens from environmental origins” explains Carmen Buchrieser, from the Biology of intracellular bacteria Unit. An interesting concept in evolution that will be applicable to other bacteria. There are many amoeba-associated bacteria in the environment, thus it is plausible that other bacterial species may evolve similar strategies and infect humans.
Source
More than 18,000 effectors in the Legionella genus genome provide multiple, independent combinations for replication in human cells, PNAS, Janvier 2019
Laura Gomez-Valero1,2, Christophe Rusniok1,2, Danielle Carson3, Sonia Mondino1,2, Ana Elena Pérez-Cobas1,2, Monica Rolando1,2, Shivani Pasricha4, Sandra Reuter5, Jasmin Demirtas1,2, Johannes Crumbach1,2, Stephane Descorps-Declere6, Elizabeth L. Hartland4,7,8, Sophie Jarraud9,10, Gordon Dougan5, Gunnar N. Schroeder3,11, Gad Frankel3, Carmen Buchrieser1,2
1 Institut Pasteur, Biologie des Bactéries Intracellulaires, Paris, France
2 CNRS UMR 3525, Paris, France
3 Medical Research Council Centre for Molecular Bacteriology and Infection, Department of Life Sciences, Imperial College London, United Kingdom
4 Department of Microbiology and Immunology, the Peter Doherty Institute for Infection and Immunity, University of Melbourne, Melbourne, Australia
5 Wellcome Trust Sanger Institute, Hinxton, Cambridge, United Kingdom
6 Institut Pasteur, Center of Bioinformatics, Biostatistics and Integrative Biology, Paris, France
7 Centre for Innate Immunity and Infectious Diseases, Hudson Institute of Medical Research, Clayton, Australia
8 Department of Molecular and Translational Science, Monash University, Clayton, Australia
9 International Center for Infectiology Research, INSERM, U1111, CNRS, UMR 5308, Université Lyon 1, Lyon, France
10 National Reference Centre of Legionella, Hospices Civils de Lyon, France
11 Centre for Experimental Medicine, Queen’s University Belfast, United Kingdom
This study is part of the priority scientific area Emerging infectious diseases of the Institut Pasteur's strategic plan for 2019-2023.