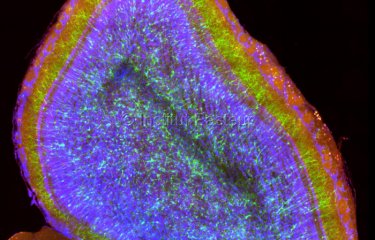
Central nervous system infections are devastating diseases, with a high mortality rate and long-term neurological disabilities in those who survive. They can be caused by a variety of pathogenic microorganisms – bacteria, fungi or viruses. One characteristic shared by all these pathogens is their ability to cross a major protective barrier in the brain, the blood-brain barrier. Scientists from the Institut Pasteur in Paris and the Hellenic Pasteur Institute in Athens have developed a new model of infection to elucidate the mechanisms used by these neurotropic pathogens to pass from the bloodstream to the brain.
The blood-brain barrier (BBB) is formed of the walls of all the blood vessels that irrigate the brain. These walls have specific filter properties, enabling them to regulate what can enter the brain. Their role acts as a double-edged sword, both protecting the brain from harmful substances but also preventing the entry of therapeutic molecules.
Billel Benmimoun is the lead author of this study and a scientist in the Brain Plasticity in Response to the Environment Unit, led by Pauline Spéder at the Institut Pasteur. He explains that "understanding how some microorganisms have acquired the ability to cross this barrier is vital, not only to identify ways to block them but also to take advantage of their strategies to deliver therapeutic substances to the brain."
A model using vinegar flies to study the BBB
The team of scientists developed an innovative model to investigate how pathogenic agents cross the BBB and their consequences for brain health. This model uses the vinegar fly (or Drosophila), a powerful genetic tool. It is unique in that it combines the integrity of the BBB structure, high analysis potential and experimental simplicity. Using the model, the scientists were able to observe and study the BBB of an intact developing brain, which proved to be sensitive to various pathogens known to cause central nervous system infections in humans, including meningococci, pneumococci and group B streptococci.
The lipoprotein Blr allows group B streptococci to cross the BBB
The model enabled the scientists to identify a new brain entry mechanism used by group B streptococci, bacteria responsible for most cases of neonatal meningitis. They identified a bacterial factor, the lipoprotein Blr, present on the surface of these bacteria. The lipoprotein binds to a receptor in the BBB and allows the bacteria to enter via endocytosis. The bacteria use the receptor as a Trojan horse to cross the barrier, hijacking its physiological role to reach the brain.
The role of Blr in brain invasion confirmed in mice
Working in collaboration with the group led by Rebecca Matsas at the Hellenic Pasteur Institute, the scientists also demonstrated that this invasion mechanism demonstrated in Drosophila is conserved in mice. In particular, the loss of Blr entirely prevents mortality from group B streptococcus infection. Given the presence of similar receptors on the human BBB, this brain entry mechanism used by group B streptococci is a promising therapeutic target.
The original model developed by the scientists represents a new gateway to understanding the cellular and molecular mechanisms that allow pathogenic agents to enter the brain. In the long term this model could pave the way for novel therapeutic approaches to block or promote access to the brain via the BBB.
Source:
An original infection model identifies host lipoprotein import as a route for blood-brain barrier crossing, Nature communications, November 30, 2020
Billel Benmimoun 1, Florentia Papastefanaki 2, Bruno Périchon 3, Katerina Segklia 2, Nicolas Roby 1,
Vivi Miriagou 4, Christine Schmitt5, Shaynoor Dramsi 3, Rebecca Matsas 2 & Pauline Spéder 1
1 Institut Pasteur, Brain Plasticity in Response to the Environment, CNRS, UMR3738 Paris, France.
2 Laboratory of Cellular and Molecular Neurobiology-Stem Cells, Department of Neurobiology, Hellenic Pasteur Institute, Athens, Greece.
3 Biology of Gram-Positive Pathogens Unit, Institut Pasteur, CNRS, UMR 2001 Paris, France.
4 Laboratory of Bacteriology, Department of Microbiology, Hellenic Pasteur Institute, Athens, Greece.
5 Ultrastructure UTechS Ultrastructural Bioimaging Platform, Institut Pasteur, Paris, France.