Ludovic Deriano’s Unit studied cells mutated for NHEJ, the canonical repair pathway for DNA double-strand break. The researchers showed that those transformed cells rely on alternative repair activities (alternative NHEJ) to survive DNA damage induced, in non-cycling (a state where the cell stops growing and dividing) G1 conditions. This alternative repair pathway represents an attractive target for future DNA repair-based cancer therapies.
Cancer cells frequently harbor somatic mutations leading to altered DNA repair pathways causing genomic instability and promoting tumor onset and evolution. The concept of synthetic lethality describes configurations where one mutation is viable for the cell but if another specific mutation occurs the combination of both mutation is lethal for the cell. This concept can be exploited in such malignancies as these tumor cells, but not healthy cells in normal tissues, which relies on compensatory alternative end joining activities to survive endogenous or cancer therapy-related genotoxic stress. In this study, the Genome integrity, Immunity and Cancer Unit (Ludovic Deriano) studied transformed cells, - mutated for the canonical DNA double-strand break repair pathway called NHEJ (for non-homologous end-joining), usually happening in G1 phase, the first of four phases of the cell cycle. They show that those transformed cells rely on alternative repair activities to survive DNA damage induced, in non-cycling (a state where the cell stops growing and dividing) G1 conditions. The choice of repair pathways of DNA double-strand breaks is dependent upon the cell cycle phases (G1, S, G2…).
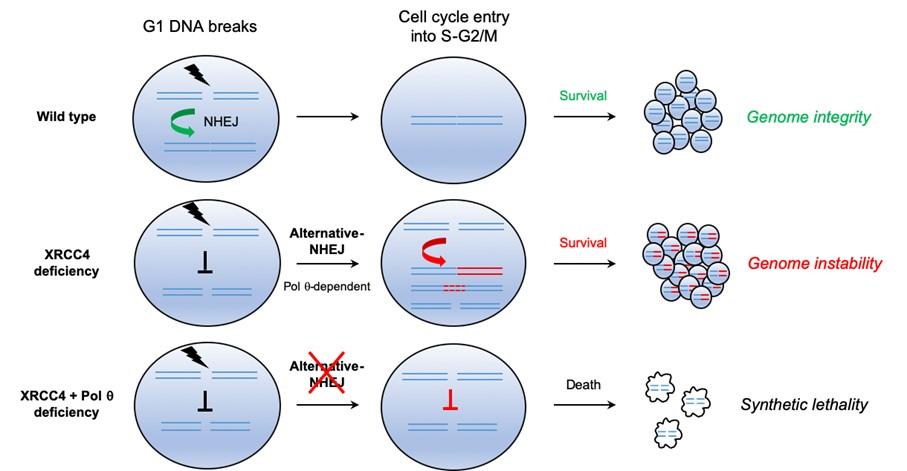
Once damaged cells resume cell proliferation, alternative repair mediated by the DNA polymerase theta (Polθ) generates genetic instability in the form of large genomic deletions and chromosomal translocations. Importantly, the study also show how cell vulnerability to G1 DNA damage can be therapeutically exploited to treat certain cancers by inhibiting the alternative NHEJ.
This study is part of the Cancer Initiative of the Institut Pasteur's strategic plan for 2019-2023.


