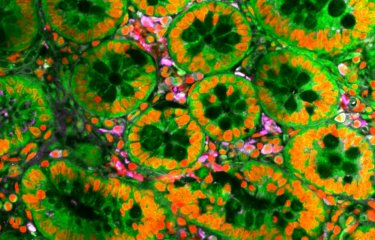
Extremophiles are organisms that have evolved to thrive under extremely harsh environmental conditions. Scientists have always been mystified by how these organisms survive in such settings and especially how extracellular appendages, such as pili, remain stable. Researchers from the Institut Pasteur, in collaboration with colleagues from the University of Virginia revealed how sugars make those pili almost indestructible. The results have been published in Nature Microbiology.
Viruses infecting extremophilic archaea (single-celled organisms) constitute an integral, yet unique part of the virosphere. The ways these viruses interact with their hosts are therefore also likely to be unique. Researchers from the Molecular Biology of Gene in Extremophiles Unit at the Institut Pasteur have found that one of their model viruses recognizes its host cell by binding to type IV pili on the cell surface. However, they were not able to identify the constituent protein of these pili by conventional techniques, such as mass spectrometry. Instead, identity of the protein was determined directly by a combination of cryo-electron microscopy and bioinformatics analyses.
A sugar protection around pili
In collaboration with a team from University of Virginia (USA), the structure of the pilus was reconstructed to 4.1 Å-resolution, explaining why the pilus was so stable and refractory to proteomic analyses. Structural data showed that it is glycosylation (sugar coating) which provides solubility to the highly hydrophobic pilus, rendering it extremely stable.
“Type IV pili are also prevalent in bacteria, including human pathogens, such as Neisseria gonorrhoeae and Pseudomonas aeruginosa. However, unlike their extremophilic counterparts, bacterial pili are not covered with such an extensive armor and would readily fall apart under extreme conditions” says Mart Krupovic, group leader in the Molecular Biology of Gene in Extremophiles Unit at the Institut Pasteur. “Extensive sugar-coating is one of many adaptations which extremophiles have evolved to survive the harshest imaginable conditions and there’s a lot we can still learn from them” he continues.
Towards indestructible coat around proteins
This data explains how macromolecular complexes remain stable under extremely harsh conditions of high temperature and acidic pH. This is a first step to harnessing these molecular solutions invented by extremophiles to improve the stability of proteins that are being used in various molecular biology, diagnostic and biomedical applications.
NB: The research was supported by the National Institutes of Health, grants GM122510 and GM123089, as well as l’Agence Nationale de la Recherche project ENVIRA, grant ANR-17-CE15-0005-01. Kreutzberger was supported by NIH grant T32 GM080186.
Source
An extensively glycosylated archaeal pilus survives extreme conditions, Nature Microbiology, May 20th, 2019
Fengbin Wang1, Virginija Cvirkaite-Krupovic2, Mark A. B. Kreutzberger1, Zhangli Su1, Guilherme A. P. de Oliveira1, Tomasz Osinski1, Nicholas Sherman3, Frank DiMaio4, Joseph S. Wall5, David Prangishvili2, Mart Krupovic2* and Edward H. Egelman1*
1 Department of Biochemistry and Molecular Genetics, University of Virginia, Charlottesville, VA, USA
2 Unité de Biologie Moléculaire du Gène chez les Extrêmophiles, Institut Pasteur, Paris, France
3 Department of Microbiology, Immunology and Cancer Biology, University of Virginia, Charlottesville, VA, USA
4 Department of Biochemistry, University of Washington, Seattle, WA, USA
5 Brookhaven National Laboratory, Upton, NY, USA
* corresponding authors.