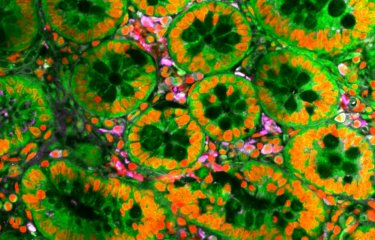
Mitochondria are organelles that provide most of the energy necessary for the cell. This energy is provided in “tokens” of adenosine triphosphate, or ATP, a molecule that is required in most chemical reactions and that is essential for all biological processes, such as locomotion, cell division, and transfer of material across membranes. Cancer cells also require energy to proliferate, and for this they need functional mitochondria. In an article published in Cell Metabolism, scientists from Europe, Asia, North America and Oceania, coordinated by Prof. Jiri Neuzil (Czech Republic and Australia), and including Institut Pasteur, reveal an unexpected role of mitochondria in the proliferation of cancer cells. With this discovery, they also identify a novel target for anti-cancer treatments.
Mitochondrial ATP is produced by a complex machinery located in this organelle, which is in part coded [which means the genetic message is “written”] in the nucleus and in part in the mitochondrion, since this organelle carries its own genome (DNA).
Other mechanisms than this also produce ATP in human, animal, and plant cells, and in particular in cancer cells - under exclusive control of the nuclear genome - but these mechanisms are less efficient. Therefore, if mitochondria are impaired, less energy is produced and cells can undergo severe alterations, sometimes leading to death.
A biochemical pathway implied in cancer proliferation
It was previously shown that cancer cells with inactive mitochondria (because they lack mitochondrial DNA), and therefore devoid of the energy produced by these organelles, do not proliferate, and the tumour does not develop until they acquire mitochondria (with DNA) from host cells. It was therefore believed that cancer cells require active mitochondria because these organelles must produce the ATP necessary for proliferation.
However, our work reveals that mitochondrial DNA, and thereby fully functional mitochondria, are necessary for tumour cell proliferation not because they produce ATP, but because mitochondria power an independent biochemical pathway that produces pyrimidines. These molecules are essential components of nucleic acids (DNA), and also have regulatory functions in the cell.
A large number of experiments were necessary to demonstrate this important new concept: “Among others, a protocol previously developed by our team at Institut Pasteur, helped to definitely demonstrate the status of mitochondrial DNA in cancer cells at various steps of the process“ explains Miria Ricchetti, the leader of the Team Stability of Nuclear and Mitochondrial DNA, at Institut Pasteur. This protocol is the subject of a patent application filed by Institut Pasteur in 2012 (WO2012123588; Method, probe and kit for DNA in situ hybridation and use thereof).
New therapeutic perspectives
There is increasing evidence that mitochondria are essential for the cell, not only for the production of ATP. Thus, this work shows that one of the numerous “extra” functions of mitochondria (and not ATP production) is essential for cancer cells proliferation. It also reveals a novel target for anti-cancer therapies that was not previously considered, i.e. blocking the pathway of pyrimidine production in mitochondria of cancer cells that is closely linked to cancer onset and progression.
Source
Reactivation of Dihydroorotate Dehydrogenase-Driven Pyrimidine Biosynthesis Restores Tumor Growth of Respiration-Deficient Cancer Cells, Cell Metabolism, November 2018.
Martina Bajzikova1,2,17 Jaromira Kovarova1,17, Ana R. Coelho1,3,17, Stepana Boukalova1,17, Sehyun Oh4,17, Katerina Rohlenova1,18, David Svec1, Sona Hubackova1, Berwini Endaya5, Kristyna Judasova1, Ayenachew Bezawork-Geleta5, Katarina Kluckova1,19, Laurent Chatre6,7, Renata Zobalova1, Anna Novakova1, Katerina Vanova1, Zuzana Ezrova1,2, Ghassan J. Maghzal8,16, Silvia Magalhaes Novais1,2, Marie Olsinova2, Linda Krobova1, Yong Jin An4, Eliska Davidova1,2, Zuzana Nahacka1, Margarita Sobol9, Teresa Cunha-Oliveira3, Cristian Sandoval-Acuna1, Hynek Strnad9, Tongchuan Zhang10, Thanh Huynh11, Teresa L. Serafim3, Pavel Hozak9, Vilma A. Sardao3, Werner J.H. Koopman12, Miria Ricchetti6,7, Paulo J. Oliveira3, Frantisek Kolar13, Mikael Kubista1, Jaroslav Truksa1, Katerina Dvorakova-Hortova1,2, Karel Pacak11, Robert Gurlich14, Roland Stocker8,16, Yaoqi Zhou10, Michael V. Berridge15, Sunghyouk Park4, Lanfeng Dong5, Jakub Rohlena1 and Jiri Neuzil1,5
1 Institute of Biotechnology, Czech Academy of Sciences, Prague-West, Czech Republic
2 Faculty of Science, Charles University, Prague, Czech Republic
3 Center for Neuroscience and Cell Biology, University of Coimbra, UC-Biotech, Biocant Park, Cantanhede, Portugal
4 College of Pharmacy, Natural Product Research Institute, Seoul National University, Seoul, Korea
5 School of Medical Science, Griffith University, Southport, Australia
6 Department of Developmental and Stem Cell Biology, Institut Pasteur, Paris, France
7 CNRS UMR 3738, Team Stability of Nuclear and Mitochondrial DNA, Paris, France
8 Victor Chang Cardiac Research Institute, Darlinghurst, Australia
9 Institute of Molecular Genetics, Czech Academy of Sciences, Prague, Czech Republic
10 Institute for Glycomics, Griffith University, Southport, Australia
11 Eunice Kennedy Shriver Child Health and Human Development, National Institutes of Health, Bethesda, USA
12 Department of Biochemistry, Radboud Institute for Molecular Life Sciences, Radboud University Medical Centre, the Netherlands
13 Institute of Physiology, Czech Academy of Sciences, Czech Republic
14 Third Faculty Hospital, Charles University, Prague, Czech Republic
15 Malaghan Institute of Medical Research, Wellington, New Zealand
16 St Vincent’s Clinical School, UNSW Medicine, University of New South Wales, Sydney, Australia
17 These authors contributed equally
18 Laboratory of Angiogenesis and Vascular Metabolism, VIB-KU Leuven Center for Cancer Biology, Department of Oncology, KU Leuven, Belgium
19 Institute of Metabolism and Systems Research, College of Medical and Dental Sciences, University of Birmingham, Birmingham, UK
This study is part of the Cancer Initiative of the Institut Pasteur's strategic plan for 2019-2023.