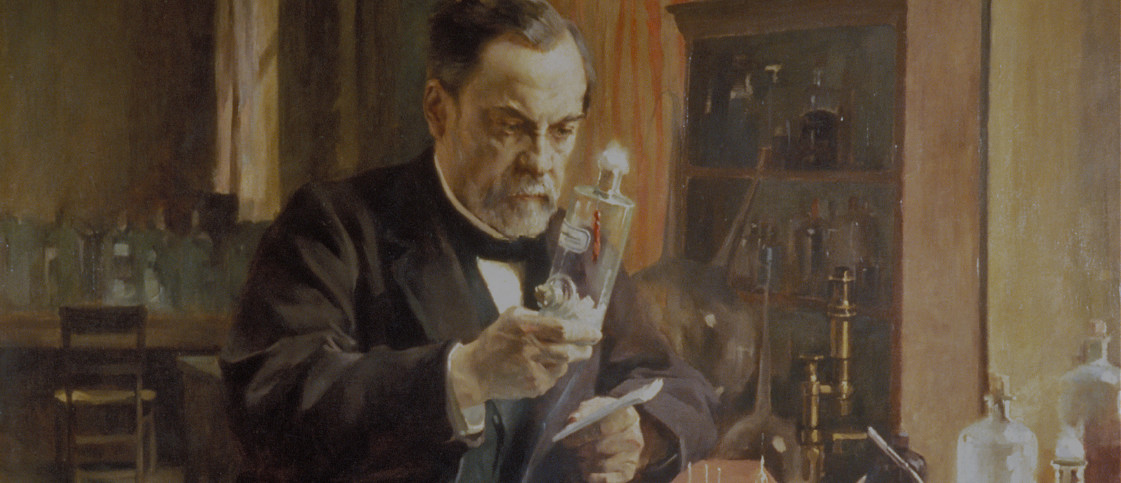Our History

"Chance favors invention only for minds prepared for discoveries by patient study and persevering efforts."
Louis Pasteur, a qualified chemist, was behind the most important scientific revolutions of the 19th century in the fields of biology, agriculture, medicine and hygiene. Beginning his research on crystallography, he soon embarked on a journey filled with discoveries which led him to develop the rabies vaccine.
Louis Pasteur’s life was filled with revolutionary discoveries and also marked by a number of events that likely fueled his desire to understand the diseases of his time. A tireless and dedicated scientist, he traveled extensively throughout France to prove his theories and solve agricultural and industrial problems caused by infectious diseases.

1822
December 27. Birth of Louis Pasteur in Dole (Jura).

1827
His family moved to Arbois.He produced his first pastel drawing at the age of 13.

1831
Louis Pasteur was a pupil at Arbois Collège

1839
Left to study at the Collège Royal in Besançon.

1840
Received his Bachelier ès lettres (Arts diploma) in Besançon. Teaching assistant at the Collège in Besançon.

1842
Received his Bachelier ès Sciences mathématiques (Mathematics diploma) in Dijon.

1843
Admitted to the prestigious École Normale Supérieure (4th place).

1845
Earned his degree in science.

1846
Appointed physics teacher at the Lycée de Tournon (Ardèche) but stayed on at the École Normale Supérieure as assistant professor.
Met Auguste Laurent in Antoine Balard’s laboratory. Studied crystals.

1847
Docteur ès sciences.
 1848
1848

Appointed physics teacher at the Lycée de Dijon then substitute chemistry teacher at the Science Faculty in Strasbourg. Research on dimorphism. Historical paper on the two forms of sodium ammonium paratartrate.
 1849
1849

Louis Pasteur married Marie Laurent, daughter of the rector of Strasbourg University. Research on the specific properties of the two constituent acids of racemic acid.
1850
Birth of his daughter Jeanne (died in 1859).

1851
Birth of his son Jean-Baptiste.Louis Pasteur’s paper on aspartic and malic acids.

1852
New research on the possibility of a relationship between crystalline form, chemical composition and the direction of rotatory polarization.
 1853
1853

Birth of his daughter Cécile (died in 1866). Louis Pasteur was named Chevalier of the Imperial Order of the Legion of Honor. He was awarded the Paris Pharmacy Society Prize for his synthesis of racemic acid. Memorandum on the discovery of the transformation of tartaric acid into racemic acid. Discovery of inactive tartaric acid.
1854
Louis Pasteur was appointed Dean of the Science Faculty in Lille.
 1855
1855

Beginning of studies on fermentation. Presentation of a paper on amyl alcohol in Lille.
 1856
1856

Beginning of research on alcohol fermentation.
 1857
1857

Appointed administrator and director of scientific studies at the École Normale Supérieure. Paper on lactic fermentation. Paper on alcohol fermentation.
 1858
1858

Birth of his daughter Marie-Louise. Set up his laboratory in the attic of the École Normale Supérieure in Rue d’Ulm, Paris. Beginning of research on spontaneous generation.
 1859
1859

Experimental Physiology Prize from the Science Academy for his work on fermentation.
 1860
1860

Air samples collected in Arbois to study spontaneous generation. Investigation of the doctrine of spontaneous generation.
 1861
1861

Awarded the Jecker Prize by the Science Academy for his research on fermentation. Publication of his findings on vinegar in the Paris Chemistry Society bulletin.
 1862
1862

Elected to the Science Academy (mineralogy section). Study of the role of mycoderma in acetic fermentation. Awarded the Alhumbert Prize for his research on spontaneous generation.
 1863
1863

Birth of his daughter Camille (died in 1865). Napoleon III requested Louis Pasteur to study wine diseases. Research on wine and the effect of oxygen in air on the winemaking process. Appointed professor of applied geology, physics and chemistry at the École des Beaux-Arts.
 1865
1865

Filed a patent for a process used to preserve and improve wine by moderate heating in anaerobic conditions, which came to be known as pasteurization. Research on silkworm diseases.
 1866
1866

Publication of "Studies on wine". Publication of an essay on the scientific work of Claude Bernard. (portrait above)

1867
Set up a physiological chemistry laboratory at the École Normale Supérieure. Appointed professor of organic chemistry at the Sorbonne. Awarded the Grand Prix at the Universal Exposition for his research on wine. Resigned from his administrative duties at the École Normale Supérieure.

1868
Degree of Doctor of Medicine honoris causa from the University of Bonn. Made a Commander of the Legion of Honor. Publication of research on vinegar. Louis Pasteur suffered a stroke that paralyzed his left side.
 1870
1870

Publication of research on silkworm diseases.
 1871
1871

Research on new processes for manufacturing and preserving beer.
 1873
1873

Elected to the Academy of Medicine.
 1876
1876

Publication of "Studies on beer".
 1877
1877

Memorandum on the fermentation of urine. Research on anthrax. Research on septicemia.
 1878
1878

Made a Grand Officer of the Legion of Honor. Publication of the paper "Germ theory and its applications to medicine and surgery". Developed the chicken cholera vaccine using an attenuated microbe. Research on gangrene, septicemia and puerperal fever.
 1879
1879

Paper on the plague. Discovery of immunization using attenuated cultures. His daughter Marie-Louise married René Vallery-Radot. (Portrait above)
 1880
1880

Appointed member of the Central Society of Veterinary Medicine. Paper on virulent diseases (Louis Pasteur established the principle of virus vaccines for the very first time). Beginning of research on rabies.
 1881
1881

Received the Grand Croix of the Legion of Honor. Developed the anthrax vaccine. Work on yellow fever near Bordeaux. Elected to the Académie française.

1882
Paper on contagious pleuropneumonia in cattle. Research on erysipelothrix infection.
 1883
1883

Developed the erysipelothrix vaccine with Louis Thuillier (Portrait above).
 1884
1884

New papers on rabies. Paper on pathogenic microbes and attenuated-virus vaccines at the Copenhagen Congress. Louis Pasteur presented the general principle of vaccination of virulent diseases.
 1885
1885

First human rabies vaccination.
 1887
1887

Elected Life Secretary of the Science Academy. Suffered a second attack of paralysis.
 1888
1888

Opening of the Institut Pasteur on November 14.
 1892
1892

Jubilee celebrated at the Sorbonne on December 27.
 1895
1895

Louis Pasteur died in Villeneuve-l’Étang on September 28. (Last photograph taken of Louis Pasteur)
"Adopt a critical mind. By itself, it can neither encourage ideas nor stimulate anything great. But without it, everything is useless. It always has the last word."
Louis Pasteur’s work
Every discovery opened a new field of investigation leading to further breakthroughs.
Louis Pasteur’s life’s work can be divided into three main parts. Chemistry and the observation of crystals led him to study fermentation.
And this led to his disproving of the spontaneous generation theory, a key discovery which opened the doors to microbiology and vaccination.


The early years 1847-1862
1847
Research on molecular asymmetry
In 1847 Louis Pasteur, a young chemist freshly graduated from the prestigious Ecole normale supérieure, set to work on the problem posed by German physicist Eilhard Mitscherlich, namely, why do sodium ammonium paratartrate and tartrate - two seemingly identical chemical substances - affect polarized light differently ?
The middle years 1862-1877
1865
deposit of the pasteurization
patent
Louis Pasteur’s work raised a new set of research questions, such as " Where do fermentation agents come from ? " and " Do they originate from germs similar to themselves or do they appear spontaneously as explained by the spontaneous generation theory ? "


The final years 1877-1887
1885
FIRST HUMAN RABIES
VACCINATION
Between the age of 55 and 65 Louis Pasteur developed microbiology, applying it to medicine and surgery. Having established that diseases were caused by microorganisms, he then sought to identify and find a means of fighting them. His finest accomplishment was rabies.
1888
CREATION OF THE INSTITUT PASTEUR
The Institut Pasteur is named after its illustrious founder and owes much to this scientific genius. Yet its story is also linked to the lives and discoveries of many other scientists, all inspired by the humanist ideals of Louis Pasteur, whose scientific breakthroughs have benefited people's health worldwide.
The Institut Pasteur is a private, non-profit foundation officially recognized for charitable status, just as Louis Pasteur himself wanted.
Established by decree on June 4, 1887, the Institut Pasteur was opened on November 14, 1888 following Louis Pasteur's successful international appeal for funds. He now had the facilities to extend vaccination against rabies, continue research on infectious diseases and share the resulting knowledge.

THE INSTITUT PASTEUR THROUGH HISTORY

A year after the Institut Pasteur opened, Emile Roux launched the world's first ever microbiology course, entitled "Technical Microbiology". Students from all over Europe came to attend theoretical and practical training in this new scientific discipline.

The first Institut Pasteur outside France was set up by Albert Calmette in Saigon in 1891. Its role was to administer vaccines for rabies and smallpox in Indochina.

Opening of the Institut Pasteur in Antananarivo, Madagascar.

A permanent mission from the Institut Pasteur in Paris, directed by the Sergent brothers, was sent to tackle malaria in Algeria. This mission merged with an anti-rabies center to create the Institut Pasteur in Algeria. In 1960, the Institut Pasteur in Algeria launched a program to combat tuberculosis.

Opening of the Institut Pasteur in Tunis.

In April 1886, sixteen Russian patients were treated for rabies by Louis Pasteur and returned to Russia cured of the disease. Pasteur's nephew Adrien Loir traveled to Saint Petersburg to set up the first anti-rabies center outside France. In 1923, on the centenary of Louis Pasteur's birth, the Pasteur Anti-Rabies Center, associated with the Imperial Institute of Experimental Medicine, became the Institut Pasteur in Saint Petersburg. It joined the Institut Pasteur International Network in 1993.

Jean Laigret developed the yellow fever vaccine at the Institut Pasteur in Dakar. Georges Girard and Jean Robin developed a plague vaccine at the Institut Pasteur in Madagascar.

When the Institut Pasteur in French Guiana was set up, its main contributions were the development of a treatment for leprosy and epidemiology research on malaria and yellow fever.

In August 1946, the Institut Pasteur in Nha Trang (Vietnam) set up a veterinary annex in Phnom Penh, Cambodia, to produce the rinderpest vaccine. This resulted in the creation of the Institut Pasteur in Phnom Penh in December 1953. It was renamed the Institut Pasteur in Cambodia in 1958 and shifted its focus to human biology and diseases.

The Institut Pasteur in New Caledonia was set up in 1954. Following a dengue outbreak which affected 40% of the population, an Institut Pasteur mission in Nouméa identified the viral type and paved the way for the creation of an arbovirology and entomology unit in 1974.

The Institut Pasteur in Bangui, which opened in 1961, is a state-approved organization in the Central African Republic. It mainly focuses on research into arboviruses (arthropod-borne viruses).

The Institut Pasteur in Côte d'Ivoire, which opened on July 27, 1972, is involved in the development of rapid diagnostic techniques for arboviruses and enteroviruses and in microbial monitoring.

In 1989, the institutes formalized their membership of the Pasteurian community, as well as their resulting rights and obligations, by adopting a General Declaration of Scientific Cooperation. The declaration was signed by each director and marked the official launch of the Institut Pasteur International Network. Ever since, despite the closure of some institutes (owing to major changes in local policy), the number of institutes has continued to grow.

The Hong Kong University – Pasteur Research Pole, created in 1999 and officially opened in 2000, focuses its research on viruses that represent a risk for public health in the region. It is currently investigating emerging and re-emerging infectious diseases in China, including SARS, influenza, AIDS, dengue and hepatitis C.

The CERMES (Center for Medical and Health Research in Niger), which joined the International Network in 2002 with the new status of associate institute, conducts research on bacterial meningitis, malaria and HIV/AIDS.

In Brussels, an institute founded in 1901 by Jules Bordet became a member of the Institut Pasteur International Network. In 2008, this institute was integrated into the Directorate of Communicable and Infectious Diseases of the Scientific Institute of Public Health (WIV-ISP), with the entire institute becoming a corresponding member of the International Network.

The Institut Pasteur in Montevideo, Uruguay, the result of trilateral cooperation between the French and Uruguayan governments and the Institut Pasteur, joined the International Network in June.

The Institut Pasteur in Laos was officially opened in Vientiane in January 2012, becoming the 32nd institute in the Institut Pasteur International Network. The aim of this new research center for infectious and parasitic diseases is to reduce the risks of pandemic outbreaks in Southeast Asia, where such diseases are rife.

On November 11, 2016, in Conakry, the President of the Republic of Guinea, His Excellency Professor Alpha Condé, laid the first stone of the Institut Pasteur in Guinea at a ceremony attended by the French Minister of Foreign Affairs and International Development, Jean-Marc Ayrault. The decision to set up an Institut Pasteur in Guinea was made in 2014 in response to the Ebola crisis. It reflects the joint determination of the French and Guinean Presidents to tackle emerging outbreaks on a long-term basis. This led to the signing of a memorandum of understanding between the Republic of Guinea and the Institut Pasteur in September 2015.
10 Nobels

1885
Development of the rabies vaccine by Louis Pasteur.
1889
Research on inflammation and immunity by Elie Metchnikoff.

1894
Development of a diphtheria treatment based on serotherapy by Emile Roux, Louis Martin and Auguste Chaillou..
1894
Identification of the plague bacillus, Yersinia pestis, by Alexandre Yersin.

1896
Elucidation of the mechanisms underpinning the immune system, especially the involvement of antibodies and the complement system, by Jules Bordet.
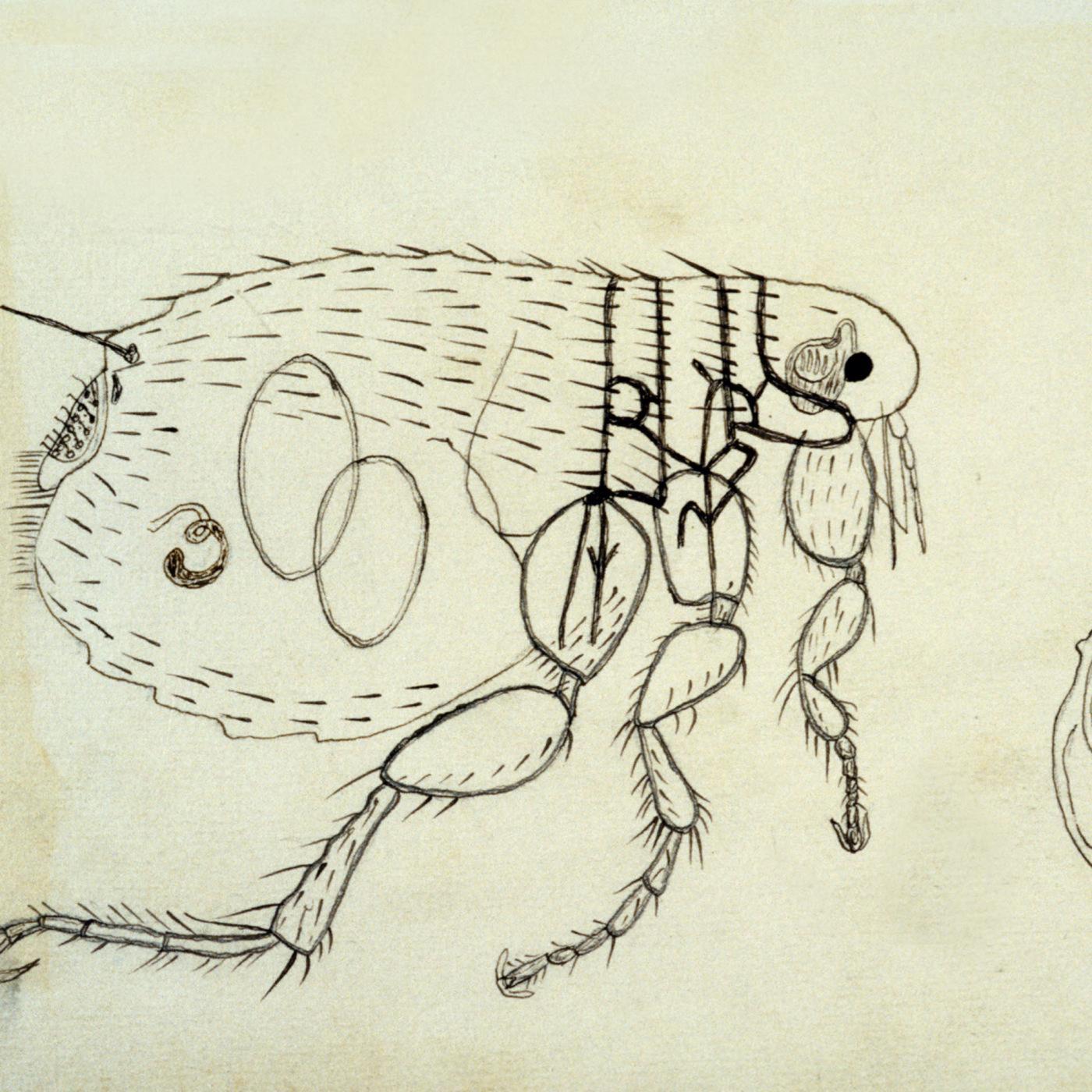
1898
Identification of the role of fleas in the transmission of plague by Paul-Louis Simond.

1917
Discovery of bacteriophages by Félix d'Hérelle. Bacteriophages are viruses that only replicate in bacteria.
1921
Development of the BCG vaccine against tuberculosis by Albert Calmette and Camille Guérin.

1932
Discovery of the action mechanisms of growth factors by André Lwoff.
1932
Development of a yellow fever vaccine by Jean Laigret.
1901
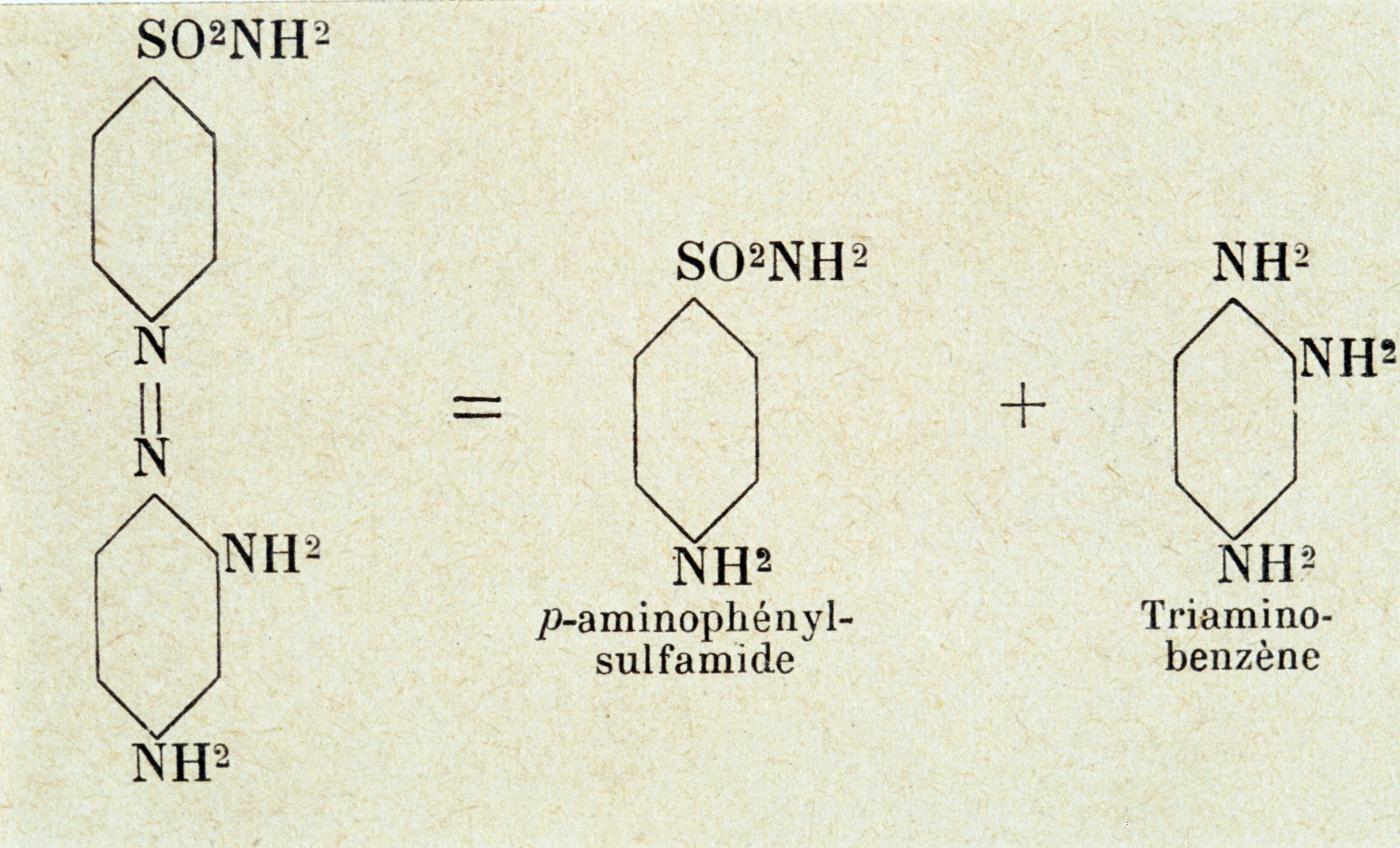
Discovery of the anti-infectious action of sulfanomides by Jacques and Thérèse Tréfouël and Federico Nitti.

1937
Discoveries relating to antihistamines and curare-like agents by Daniel Bovet.
1954
Development of a polio vaccine by Pierre Lépine.
1960
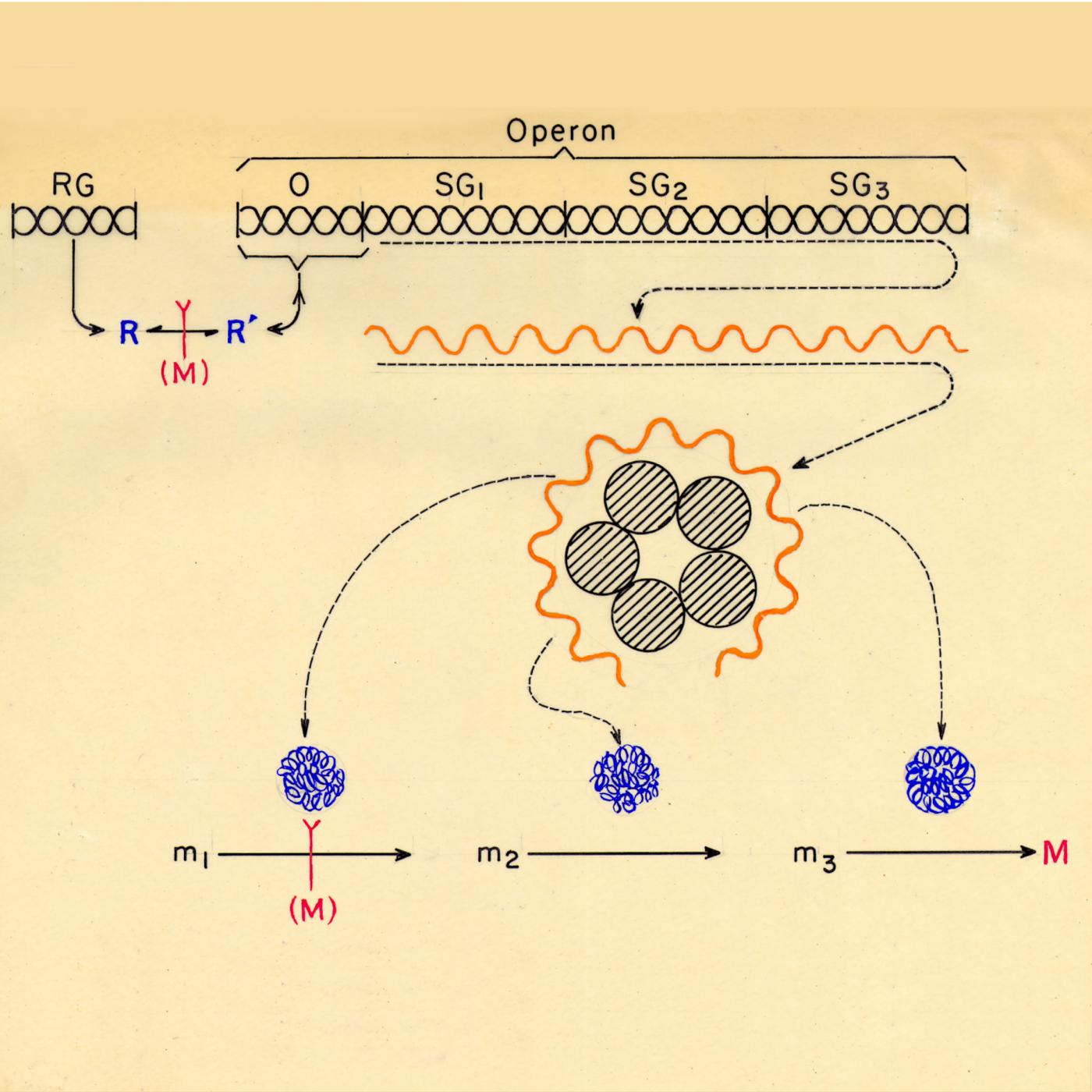
Protein biosynthesis. Research on the regulation of enzyme synthesis and activity and the mechanisms of protein biosynthesis by Jacques Monod and François Jacob.
1970
Isolation of the first neurotransmitter receptor, the acetylcholine receptor, by Jean-Pierre Changeux. Acetylcholine is a chemical messenger that transmits nerve impulses between nerve cells in the brain and between nerve cells and muscle fibers.
1983
Discovery of the AIDS viruses, HIV1 then HIV2, by Luc Montagnier, Jean-Claude Chermann and Françoise Barré-Sinoussi.
1985
Development of the hepatitis B vaccine by P. Tiollais. The hepatitis B vaccine was the first human vaccine obtained by genetic engineering from animal cells.
1991
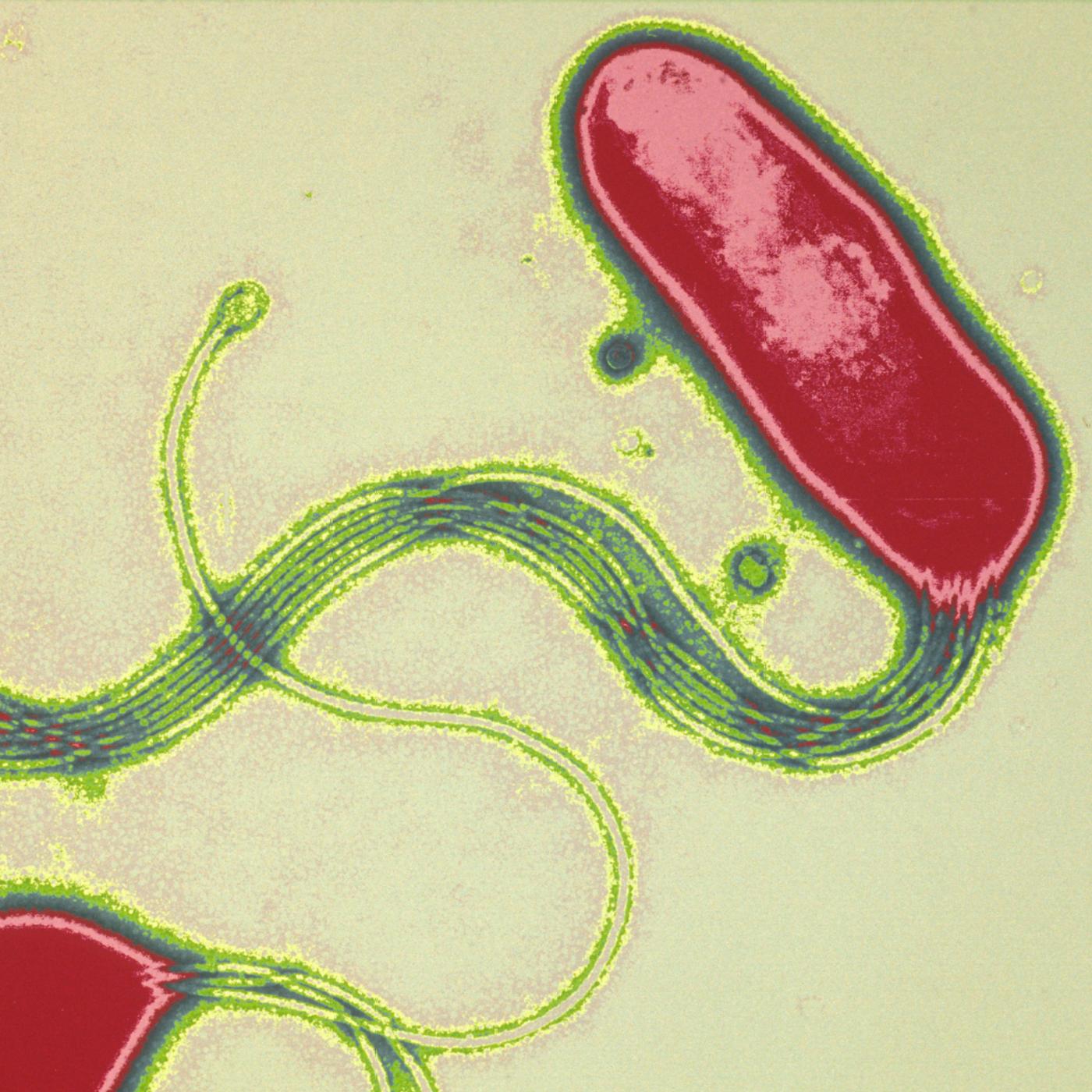
Development of a rapid diagnostic test for Helicobacter pylori bacteria by Agnès Labigne. Helicobacter pylori bacteria are responsible for stomach ulcers and cancer.

1995
Identification of genes responsible for deafness by Christine Petit.
1996
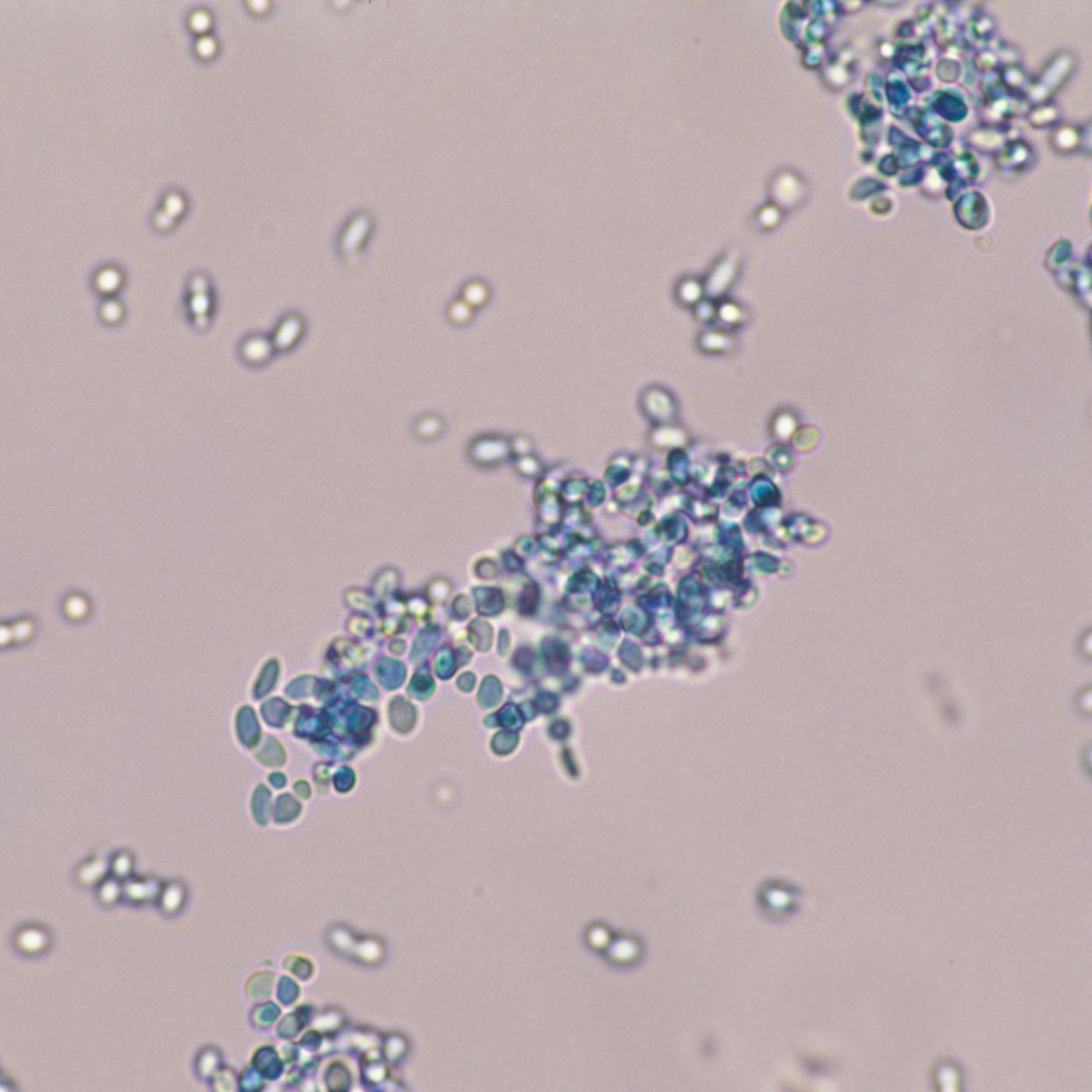
Full sequencing of the genome of the yeast Saccharomyces cerevisiae by Bernard Dujon.

1997
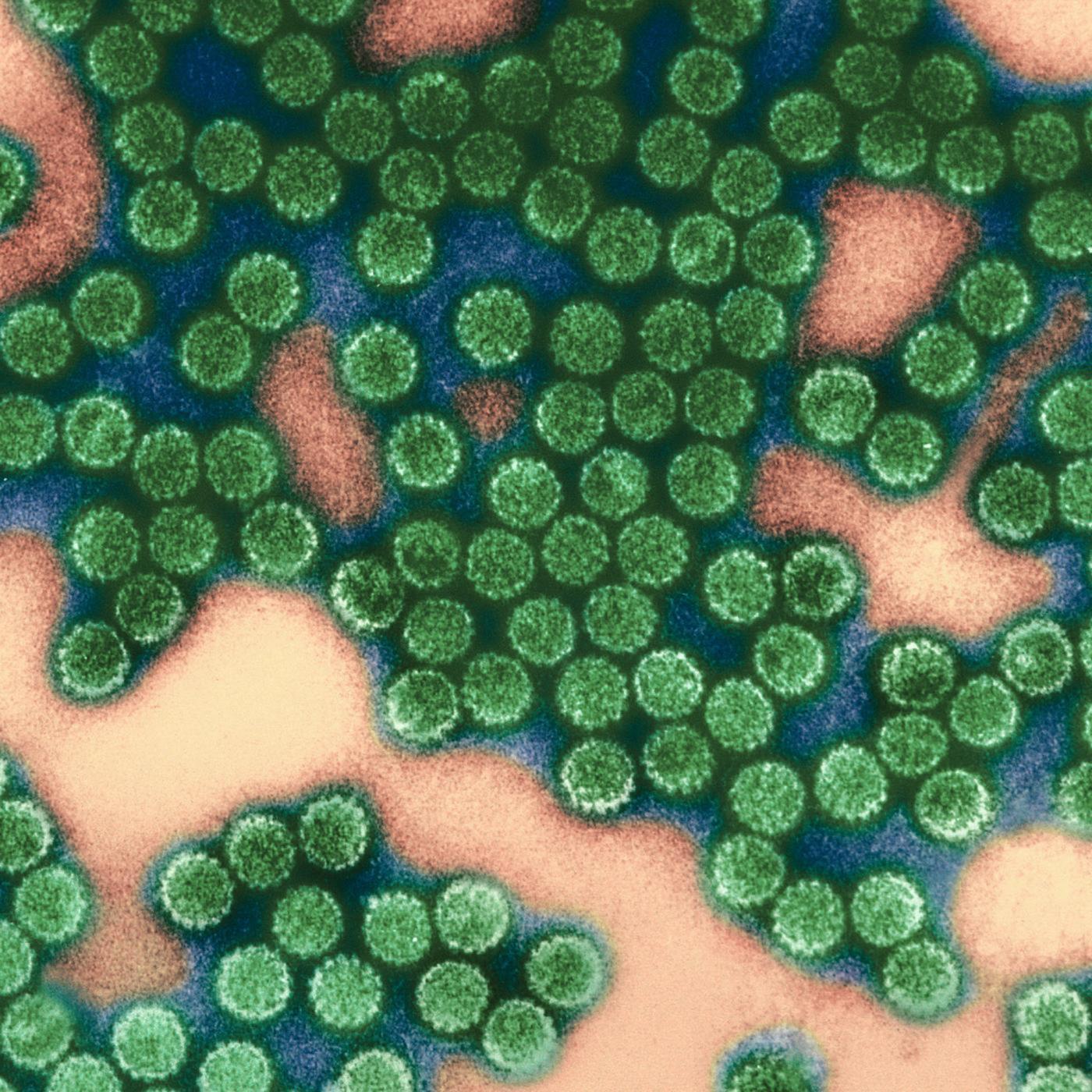
Discovery of a nicotine dependence gene by Jean-Pierre Changeux.
1998
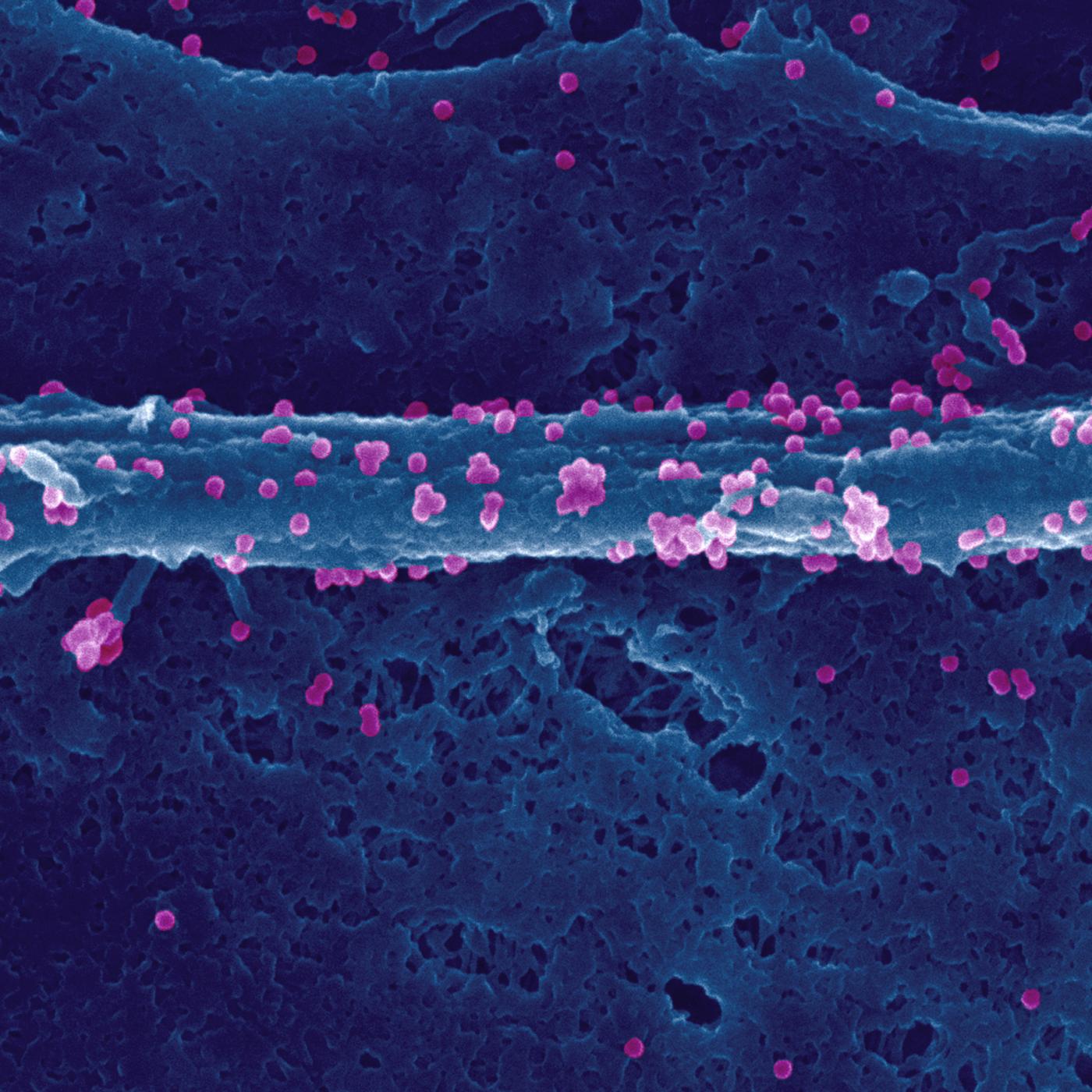
First discovery of a gene predisposing to papillomavirus infection in humans by Gérard Orth. Investigation of a potential link with psoriasis.
2006
Identification of the source of the chikungunya outbreak in the Indian Ocean by sequencing the genomes of viral strains.

2007
Explanation of the mechanism that enables the immune systems of "HIV controllers" – HIV-infected patients who do not go on to develop the disease – to control the AIDS virus. Gianfranco Pancino et al.
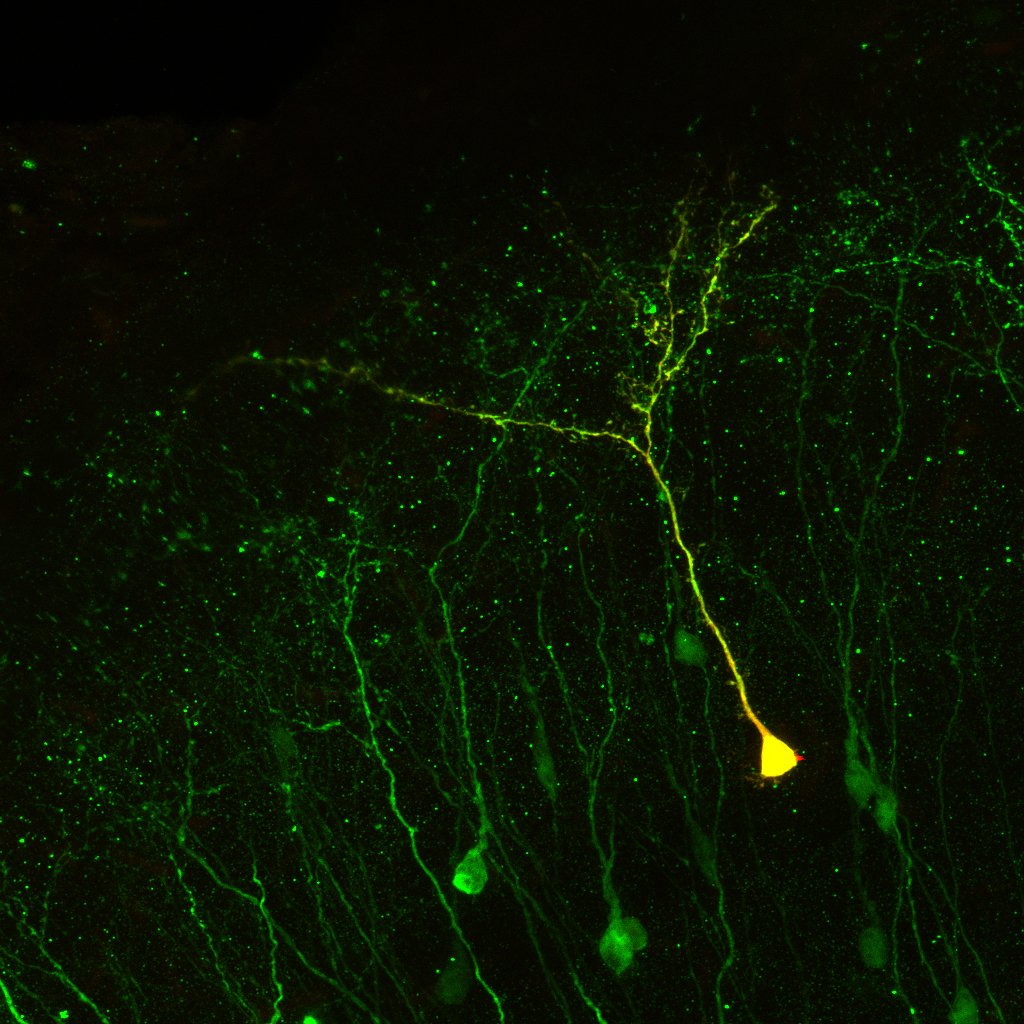
2008
A new source of neurons is identified in the adult brain. Alonso et al.
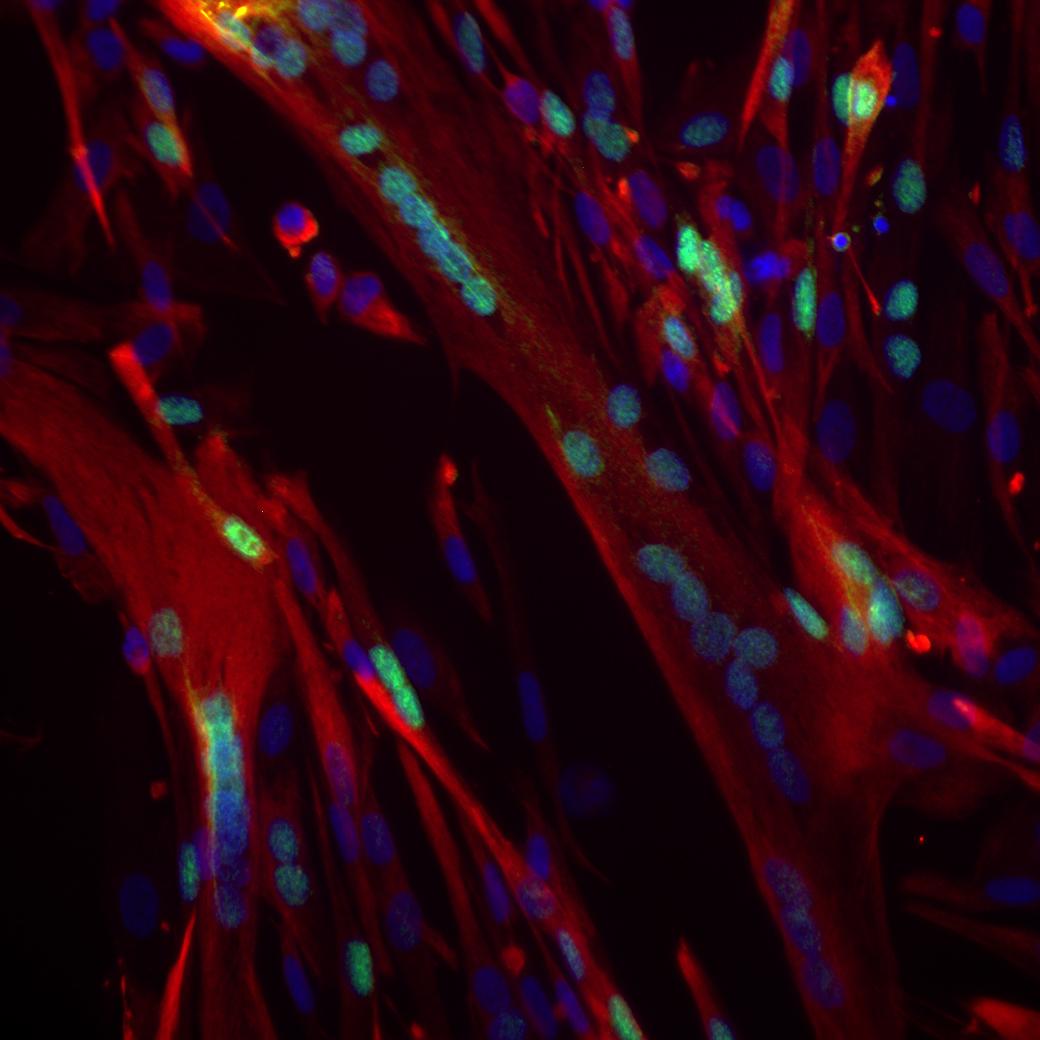
2010
The origins of hematopoietic stem cells (HSCs), which give rise to all the body's immune and blood cells, are identified by Karima Kissa and Philippe Herbomel. HSCs are formed from cells in the embryo's aortic wall.
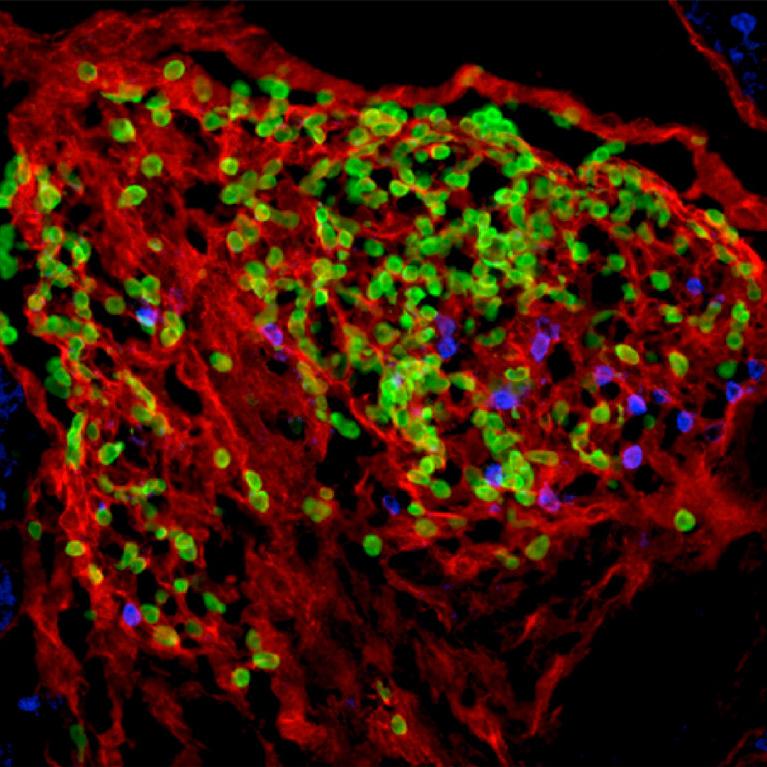
2011
Fetuses spontaneously develop an immune defense system "in advance". Sawa et al.

2012
Muscle stem cells survive seven days post mortem. This discovery paves the way for a new source of stem cells, and more importantly for new methods of preserving these cells that are used to treat a number of diseases.

2013
AIDS: 14 adult patients manage to control their HIV infection more than seven years after stopping treatment. Sáez-Cirión et al.
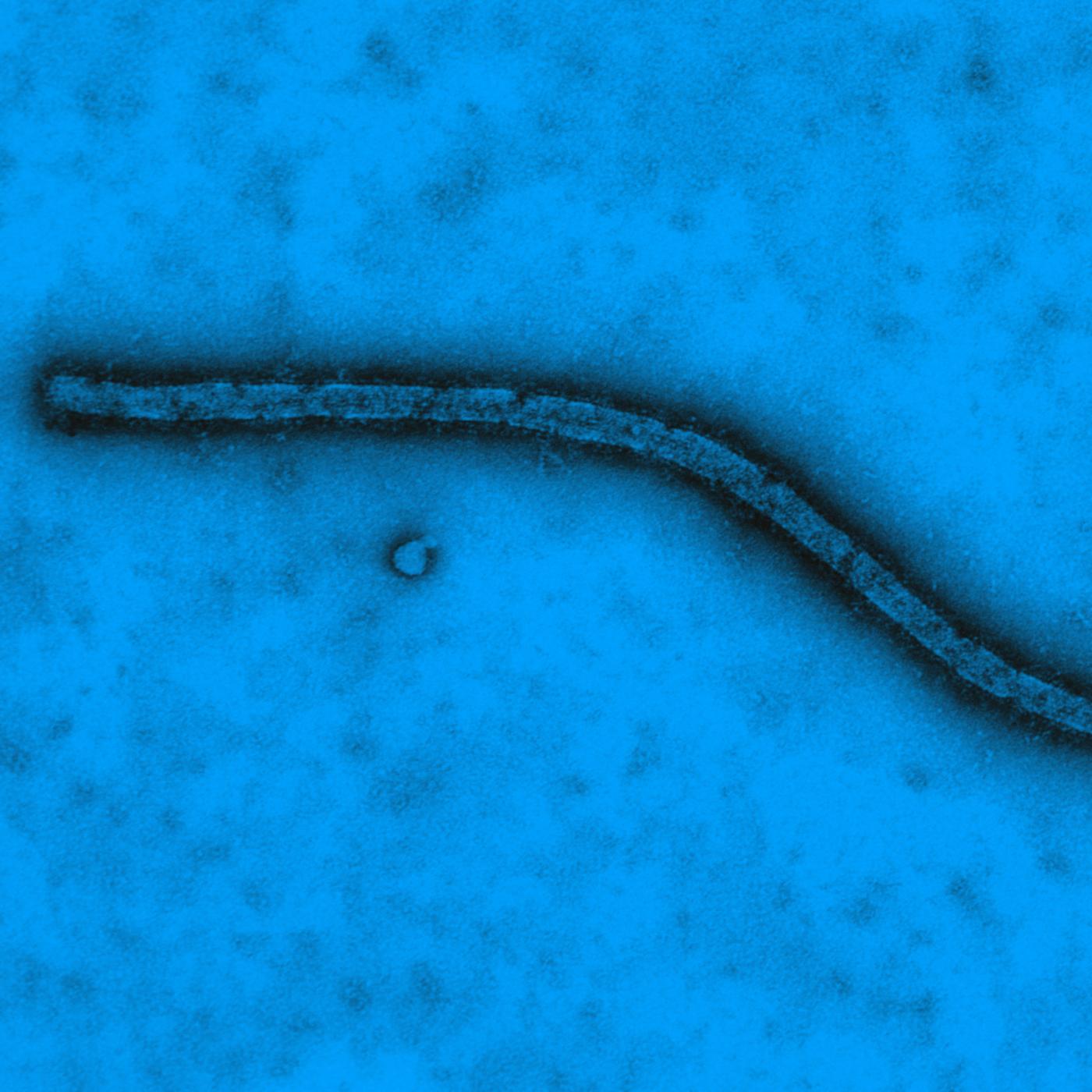
2014
Identification of a new variant of the Ebola virus in Guinea.

2015
First case of prolonged remission – twelve years – in an HIV-infected child. Sáez-Cirión et al.

2016
Publication of the first whole genome sequence of the Zika virus circulating in the Americas.